Interview to Dr Liz McCarthy
Microscopy Technical Specialist at Brunel University of London, United Kingdom
Could you tell us about your scientific background and your experience in microscopy?
I am a biologist by trade, which makes a world of ever complicated physics a little intimidating! I have always been interested in host-pathogen interactions and immunology in general. I completed my PhD at the Institute of Animal Health in Pirbright where I learned a love of microscopy. After this I moved on to an imaging technician role at the London School of Hygiene and Tropical Medicine supporting the researchers with their widefield and confocal microscopy. The role broadened and developed over my time there into Microscopy Manager and CL2 Malaria Lab Manager. Whilst I loved the team I worked with; I felt my microscopy time was being squeezed by the other demands of the role. So last year I moved on to my current position as Microscopy Technical Specialist at Brunel University of London (BUL).
What is your current role at Brunel University, and how is the facility organized?
I am the Microscopy Technical Specialist at BUL, responsible for the management, maintenance and training in our high-end light microscopy facilities. This is a relatively small facility with around 10 instruments split between 2 separate labs. The main microscopy suite sits in the second floor of the Heinz Wolff Building at our Uxbridge campus. Pop down one floor and you will find an unassuming office with the staff dedicated to supporting the light microscopy at the University, me. We have electron microscopy facilities also, these are in our Bragg building and are looked after by a dedicated team of staff within the Experimental Techniques Centre.
I work within (and share an office with) a wider team of technicians who facilitate the laboratory teaching of both undergrad and postgrad students, as well as the smooth running of the research facilities and other equipment within the College. It is a fantastic team to work with, all of whom have a wide and varied breadth of knowledge and we all have a proactive presence in TechNet (The Brunel Technician Network), which formalises and champions the Universities ongoing support as signatories to the Technician Commitment.
When it came time to purchase a new microscope, what led you to choose CrestOptics systems?
Our CrestOptics-Nikon system was purchased before I started working at Brunel; I was just lucky enough to inherit it. As a small facility we cannot always justify having multiple single-imaging technique systems – with our user base it wouldn’t be financially or environmentally sustainable. We support research centres and facilities with a variety of microscopy needs to cover areas such as anti-microbial resistance; ageing; inflammation; cancer, genome engineering and maintenance and more. Using standard cell cultures as well as organ-on-a-chip and zebrafish. As such, a multimodal system covering super resolution through to confocal and widefield microscopy was the right choice for us at the time, particularly with the accessible price point. Of course, now it is so popular we could do with another one!
“Our multimodal system, combining spinning disk and DeepSIM, has become so popular that we could easily do with another one!”
Can you describe your current microscope setup?
Affectionately termed, “The Beast”, our DeepSIM unit and X-Light V3 spinning disk are combined with a Nikon Ti2 inverted microscopy stand fitted with Prior Scientific NanoScan SP600 piezo insert. Environmental controls for CO2 and temperature support longer term live cell imaging with an OKOLAB incubator. 2 Teledyne Photometrics Kinetix cameras backed up with our triggering unit allow us to maximise the speed of acquisition for dual colour imaging of highly dynamic processes. We utilise the Nikon D-LEDI fluorescence LED illumination system for widefield imaging modality and the 89 North LDI multi-wavelength laser diode for X-Light V3 spinning disk and DeepSIM imaging.

Microscope set-up are located in the Brunel University of London microscopy facility.
Which features of the X-Light V3 spinning disk are particularly helpful for your key users’ research?
The speed of the spinning disk, in particular with the dual cameras makes this system versatile for live cell experiments, this is proving invaluable in some of our microfluidic experiments (Figure 1). The large field of view has been of particular use for those imaging larger samples (Figures 2, 3) such as zebrafish and organoids or for when high cell counts are needed at higher magnifications either for screening rare events or for boosting analysis numbers. Reducing the imaging time needed on a system in high demand is good for the users and for facility management. We are also embracing the ease in which we can switch between the different imaging modalities and using the Nikon module JOBS to automate this for more complex experiments.
Figure 1: Live-cell time-lapse of cancer cell transmigration.
Video of cancer cell transmigration across primary endothelial HUVECs. Live-cell time-lapse (large image 1×5, 20x PhFLuo) of fluorescently green CMFDA labelled-cancer cells migration and trasmigration on and across primary endothelial cells HUVEC confluent monolayer (phase contrast) over time (1 frame/15 mins, 12h). Scale bar, 200 microns. Image courtesy of Dr. Camilla Cerrutti.
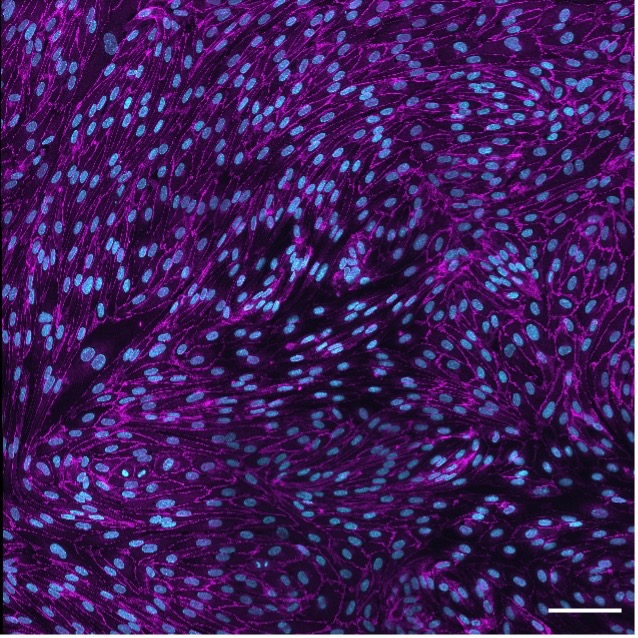
Figure 2. 2D Junctions 100 micron.
Endothelial VEcadherin junctions. Max projection of a z-stack (four stitched field of views, 40x air) of the endothelial junctional protein VEcadherin (magenta) expression by primary endothelial cells HUVEC confluent monolayer and nuclei (cyan). Scale bar, 100 microns. Image courtesy of Dr. Camilla Cerrutti.
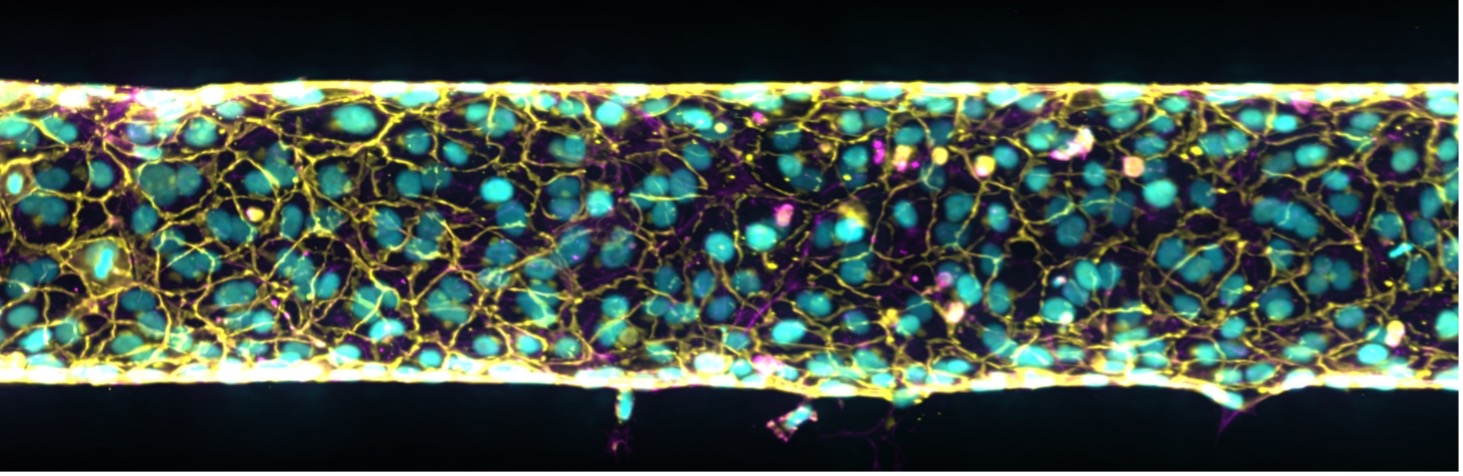
Figure 3: 3D junctions.
Endothelial VEcadherin junctions and phalloidin. Max projection of a z-stack (large image 1×5, 40x dipping) of the endothelial junctional protein VEcadherin (yellow) expression by primary endothelial cells HUVEC confluent monolayer, nuclei (cyan) and phalloidin (magenta). Image courtesy of Dr. Camilla Cerrutti.
How important is the DeepSIM super-resolution system in your key users’ work, and what are its main applications?
We have 3 micro lens arrays allowing us to optimise the DeepSIM for a variety of sample thicknesses, from single cells to organs-on-chips and organoids (Figure 4). We have also found using the DeepSIM to improve the resolution of imaging with the 20x highly beneficial. The improvement achieved negates the need for tile scanning the same area with a higher-powered objective – reducing the number of images needed, saving on data storage and user time.
Figure 4: 3D dendritic cell movie.
The movie shows a DeepSIM 3D render of a fixed dendritic cell stained with phalloidin to label actin embedded in a collagen gel. The experimental approach was used to investigate whether the actin architecture of dendritic cells was affected by Galectin-9 perturbation, and is included in the manuscript “Galectin-9 regulates dendritic cell polarity and uropod contraction by modulating RhoA activity” which has been accepted for publication at the Journal of Cell Biology. Image courtesy of Dr. Joseph Hetmanski.
Which biological applications benefit the most from the multimodal imaging platform combining X-Light V3 and DeepSIM?
From a facility management point of view the greatest benefit is the ease of the transition between standard widefield to spinning disk and to getting that greater detail from the DeepSIM. No special dyes required and no additional sample preparation needed. As long as I have done my job properly and the system is properly calibrated (which is very simple to do) then the user doesn’t need to undergo lots of extra training or go back to the lab to prep new samples if they want to step up a level. They just choose a DeepSIM optical configuration and get the software to reconstruct after acquisition. This has helped draw in those who would generally rely purely on our other widefield systems, opening up the accessibility of asking more questions of their samples. The same is true for our external users/collaborators. Being so close to London, combined with easy access by car M25/M40 has made it very easy to arrange relatively last-minute sessions. The joy on our users’ faces when they realise how easy it is to switch modalities and how good the images are, makes this work incredibly rewarding..
How do you see the future of fluorescence microscopy evolving to meet the demands of innovative biological research?
Increased demands on researchers to maximise the results in every paper with tight deadlines mean that we have to embrace the help of automated imaging and downstream analysis (Nikon’s JOBS is earning it’s keep here) with feedback microscopy and the integration of AI within these pipelines. There’s no other way to keep up. It does worry me though, as we have to make assumptions to make these algorithms work, and if we get our assumptions wrong, we could be missing out on some really interesting results without personally reviewing the biology. The amount of data we are creating in this way is huge. Data storage and general management is a constant battle for our users and I hear other facilities struggling with this also, both financially and practically. We need to play some catch up here.
What I would actually like to see is the longer-term evolution of more sustainable systems and upgrade processes. Modular systems such as the X-Light V3 light and the DeepSIM are a great example of this. The constant evolution of imaging technology and techniques and the constant drive to do more is having a huge environmental impact with equipment turnover. The scientific community is not exempt from environmental responsibilities; researchers, facilities and suppliers need to find ways to work together to improve the sustainability of our work.
“The joy on our users’ faces when they realise how easy it is to switch modalities and how good the images are, makes this work incredibly rewarding”
Acknowledgement
We would like to thank Nikon UK, Branch of Nikon Europe BV, with special thanks to Dr. Robert Tetley and Dr. Michelangelo Colavita, Advanced Imaging Specialists at Nikon, for their outstanding support and collaboration on this project at Brunel University London. We would also like to thank Vinay Vishwakarma, Microscopy Engineer, for his valuable time dedicated to the set-up.