Interview to Dr. Carolina Chavez (Facility Manager) and Dr. Alita Soch (Facility Support Officer)
The Florey – Microscopy Facility, Melbourne, Australia
Introduction of The Florey and the Microscopy Facility
The Florey is focused on cutting-edge neuroscience and mental health research. Together, the researchers aim to decipher complex medical conditions in order to uncover and build on existing knowledge. The goal of the research at The Florey is to be translational, moving from the lab bench through to patient care and ultimately new treatments. This approach ensures that fundamental discoveries have a clear path toward real-world impact, improving outcomes for patients and communities.
The Microscopy Facility offers a range of workhorse optical imaging instruments, ranging from widefield to spinning disk confocal and point-scanning confocal microscopes. Our dedicated facility team train, educate, and assist users on microscope operation, best practices, and in selecting the imaging system best suited for their experiments. The Microscopy Facility also welcomes external partners; we are an open facility supporting all researchers with their imaging experiments and encouraging scientific exchange within the wider community.
Application Overview: Imaging Workflows and Research Priorities at the Facility
Imaging experiments at the facility are highly diverse, reflecting the broad scientific questions pursued by our users. Common sample types include fluorescently labeled fixed brain tissue, organoids, neuronal rosettes, cultured cells, and zebrafish, each requiring specific imaging strategies. Many researchers in our facility rely on high-throughput volume imaging and large tile scans, which makes the spinning disk confocal system an essential approach for fast, gentle, and efficient data acquisition.
Our users increasingly value flexible workflows that allow them to move seamlessly from rapid volumetric imaging to high-resolution structural analysis. In this context, the combination of the X-Light V3 spinning disk and DeepSIM super-resolution offers a powerful multimodal imaging pipeline. The X-Light V3 enables fast, low-phototoxicity screening of large samples, while DeepSIM provides enhanced resolution for zooming in on fine subcellular features without switching platforms. This integrated multimodal approach streamlines data acquisition, improves experimental efficiency, and supports both exploratory imaging and detailed structural investigations within the same session.
“The integrated multimodal approach with X-Light V3 and DeepSIM streamlines data acquisition, improves experimental efficiency, and supports both exploratory imaging and detailed structural investigations within the same session.”
Microscope Configuration Integrating CrestOptics Technologies
Our imaging system incorporates the CrestOptics X-Light V3 spinning disk confocal, integrated with a Nikon Ti2-E microscope stand featuring a large field of view. The setup includes objectives optimized for thick sample preparations and for imaging across a broad spectral range, from 405 nm to near-IR. A compact and powerful LDI-5 laser light source from 89North provides excitation light. Two high-performance Teledyne Photometrics kinetics sCMOS cameras support rapid and sensitive data acquisition. Finally, the DeepSIM X-Light super-resolution module further enhances resolution of the system for detailed structural imaging.
Together, these components deliver a versatile and efficient imaging platform, enabling researchers to generate large volumes of high-quality data at accelerated speeds while maintaining exceptional flexibility across diverse sample types and fluorophore combinations.

Microscope set-up are located in the Microscopy Facility at The Florey.
“Together, these microscopy components create a versatile imaging platform that delivers high-quality data quickly while offering exceptional flexibility”
Case Studies from Research Users
The imaging capabilities of the system are showcased through case studies from two of our experienced users, each employing high-throughput volumetric imaging to address distinct scientific questions in their respective research fields.
a) Montanna Waters — PhD Candidate, The Florey
Research Focus: Developmental and Epileptic Encephalopathies
Montanna Waters, PhD candidate at The Florey, focuses her research on patient-derived induced pluripotent stem cells (iPSCs) to study developmental and epileptic encephalopathies (DEE), a group of severe childhood-onset neurological disorders. To model early brain development, she cultures iPSCs in micropatterned neural rosette plates, which provide a highly controlled environment resembling the earliest stages of the developing brain. By comparing neural rosettes derived from healthy controls with those generated from patients carrying epilepsy-associated genetic variants, she investigates how these mutations alter critical developmental processes such as lumen formation, cell arrangement, and neural progenitor identity.
Microscopy plays a central role in her research, as neural rosettes are complex 3D structures that cannot be fully characterized in two dimensions. Montanna uses the X-Light V3 spinning disk confocal to acquire full z-stacks across entire rosettes, allowing for detailed morphological analysis of both rosette and lumen architecture (Figure 1). The large field of view offered by the X-Light V3 enables her to efficiently image hundreds to thousands of rosettes per plate, while the confocal capabilities allow imaging deep within the tissue with high clarity.
This imaging system has been essential for generating quantitative datasets that reveal how epilepsy-associated variants disrupt early brain development. Beyond the analytical insights, the high-resolution images also showcase the elegance and complexity of human neural development in the dish. Through the integration of patient-derived stem cell models and advanced microscopy, her work aims to shed light on the cellular origins of epileptic encephalopathies and open new avenues for understanding and treating these devastating disorders.
“Combining patient-derived stem cell models with advanced microscopy, Dr. Waters research reveals the cellular origins of epileptic encephalopathies and points toward new therapeutic possibilities.”
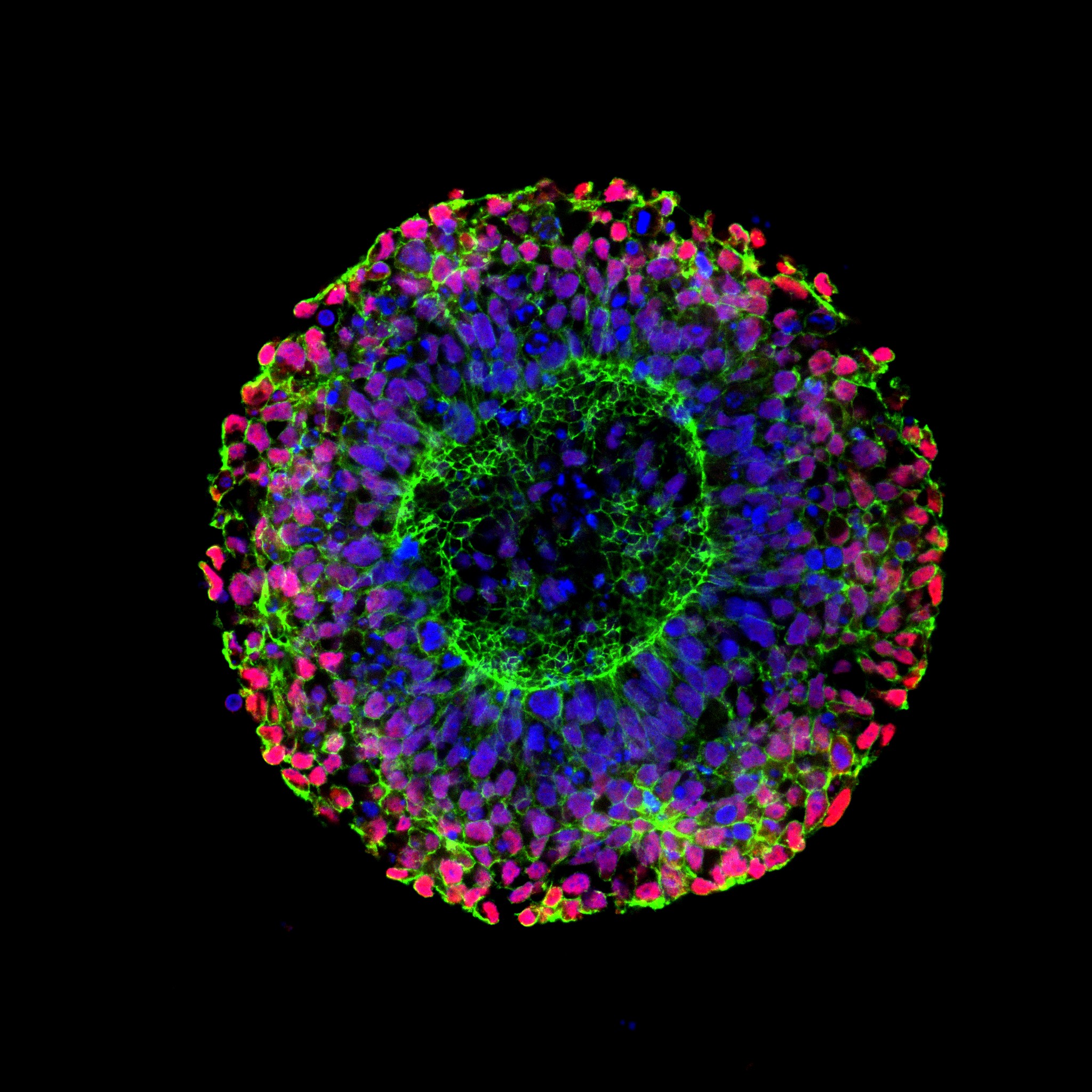
Figure 1. Stained healthy iPSC-derived neural rosette showing nuclei (DAPI, blue), cell adhesion protein (N-Cadherin, green) and neural progenitors (PAX6, red). Imaged with the 40x/1.25 silicone objective. Representative single slice of the Z-stack.
b) Lindsea Booth — Head of the Neurocardiovascular Physiology Group, The Florey
Research Focus: Neuroinflammation After Cardiopulmonary Bypass
Lindsea Booth, Head of the Neurocardiovascular Physiology Group at The Florey, investigates neuroinflammation following cardiopulmonary bypass surgery, a procedure undergone by approximately two million people worldwide each year. Although life-saving, the surgery is associated with post-operative cognitive dysfunction in nearly half of all patients. While the underlying mechanisms remain unclear, evidence suggests that neuroinflammation, hypoxia, and disruption of the blood–brain barrier are major contributing factors.
Within the Neurocardiovascular Physiology Group at The Florey, University of Melbourne, Lindsea uses the CrestOptics microscope to study neuroinflammation in preclinical models. Tissue sections from the frontal cortex are fluorescently labeled to identify microglia (IBA1), astrocytes (GFAP), and nuclei (DAPI) (Figure 2). Using either the 20x/0.8 or 40x/1.25 silicone-immersion objectives, she acquires 20–30 µm z-stacks and reconstructs the resulting 3D volumes for detailed cellular analysis.
These 3D datasets allow her and the team to assess morphological changes in microglia and astrocytes over time, providing valuable insight into how cardiopulmonary bypass triggers neuroinflammatory processes. In future work, they plan to apply these imaging approaches to assess the effectiveness of novel therapies in preventing or reversing neuroinflammation after cardiopulmonary bypass.
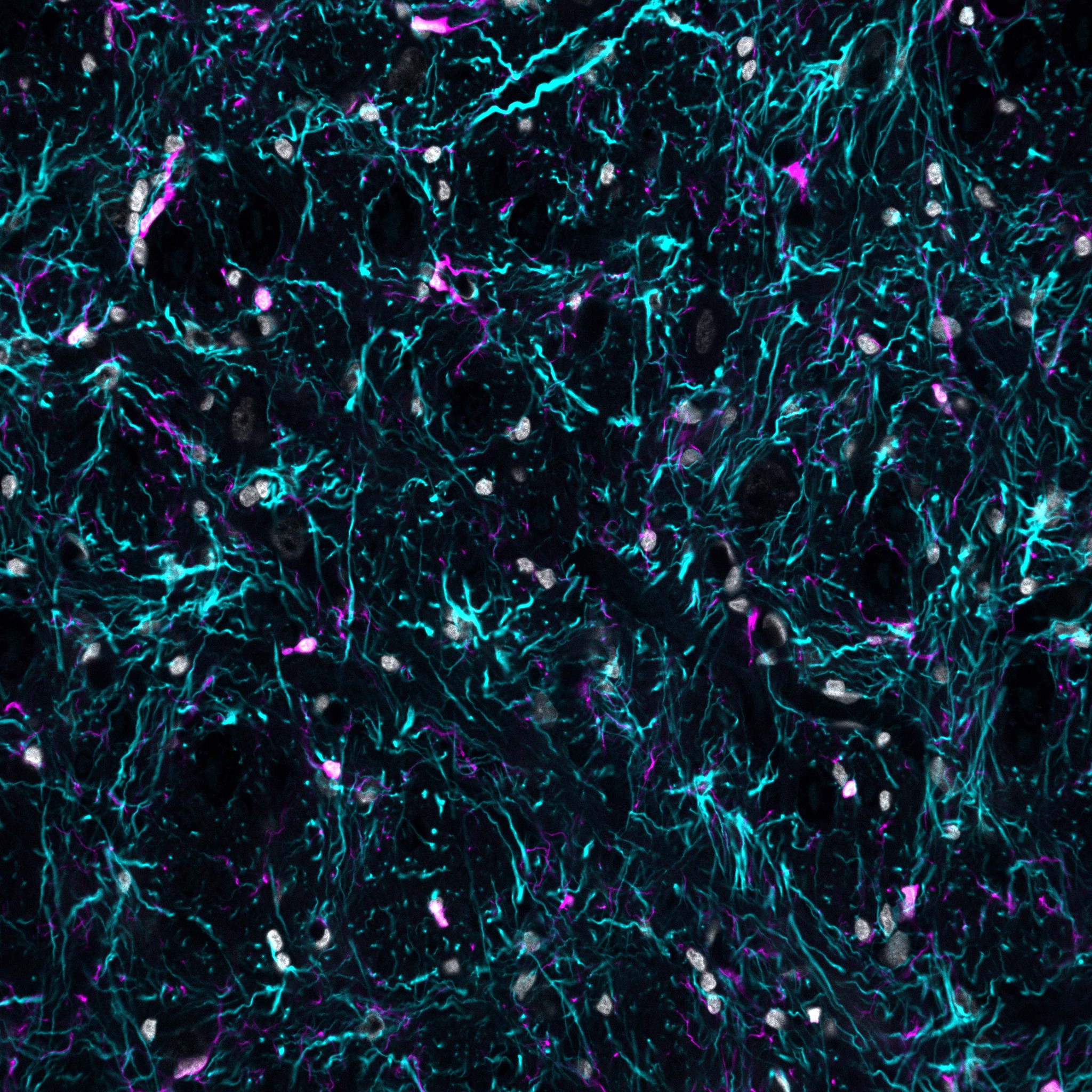
Figure 2: Stained ovine frontal lobe grey matter showing nuclei (DAPI, grey), astrocytes (GFAP, cyan) and microglia (Iba-1, magenta). Imaged with the 40x/1.25 silicone objective.
“The 3D microscopy datasets reveal how microglia and astrocytes change over time, shedding light on the neuroinflammatory impact of cardiopulmonary bypass”
CrestOptics & Klein Scientific: successful story across Australia

Klein Scientific was established by Ben Hibbs in 2020 with a view to providing researchers in Australia and New Zealand access to the latest in microscope systems, reagents and accessories.
Working closely with CrestOptics since 2022, Klein Scientific and CrestOptics now enjoy a close collaboration, with installations at leading institutions such as the University of Queensland, the University of Sydney, the University of Auckland, the University of Tasmania – and of course, the Florey.
A key focus of Klein Scientific’s work across the region is engaging closely with researchers to provide tailored microscopy support and training. As research is diverse and relies on pushing the boundaries of knowledge, Klein Scientific emphasises the importance of tailoring microscope systems to the research application. CrestOptics systems are highly flexible and can accomodate the needs of many modern research imaging requirements. Alongside a broad range of complementary and cutting-edge suppliers, Klein Scientific seeks to support researchers across Australia and New Zealand.
Ben Hibbs