Brillouin Light Scattering
Light-based Sensing
The Optical Touch on Life: revealing biomechanical frameworks through 3D Brillouin Mapping
Brillouin Microscopy is a fully optical technique that measures the 3D mechanical properties of biological samples at sub-micrometer resolution, without requiring physical contact. Unlike traditional probing methods, which are mainly contact-based and perturbative, Brillouin Microscopy can reveal local biomechanical heterogeneities at the sub-cellular level in live cells and tissues. Brillouin Microscopy enables the mapping of intracellular stiffness and viscosity in an entirely label-free and non-invasive way.
How does Brillouin Light Scattering work?
The Mechanism
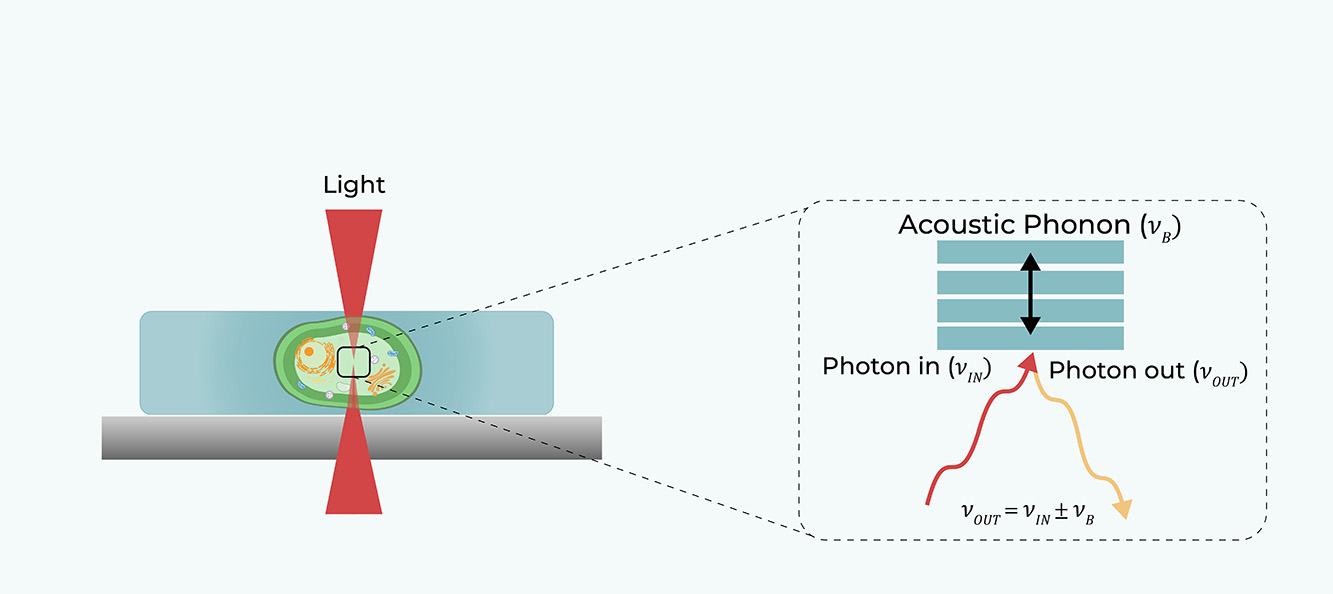
Brillouin Light Scattering is the inelastic scattering of light (photons) from density (sound) waves naturally propagating in the material (acoustic phonons).
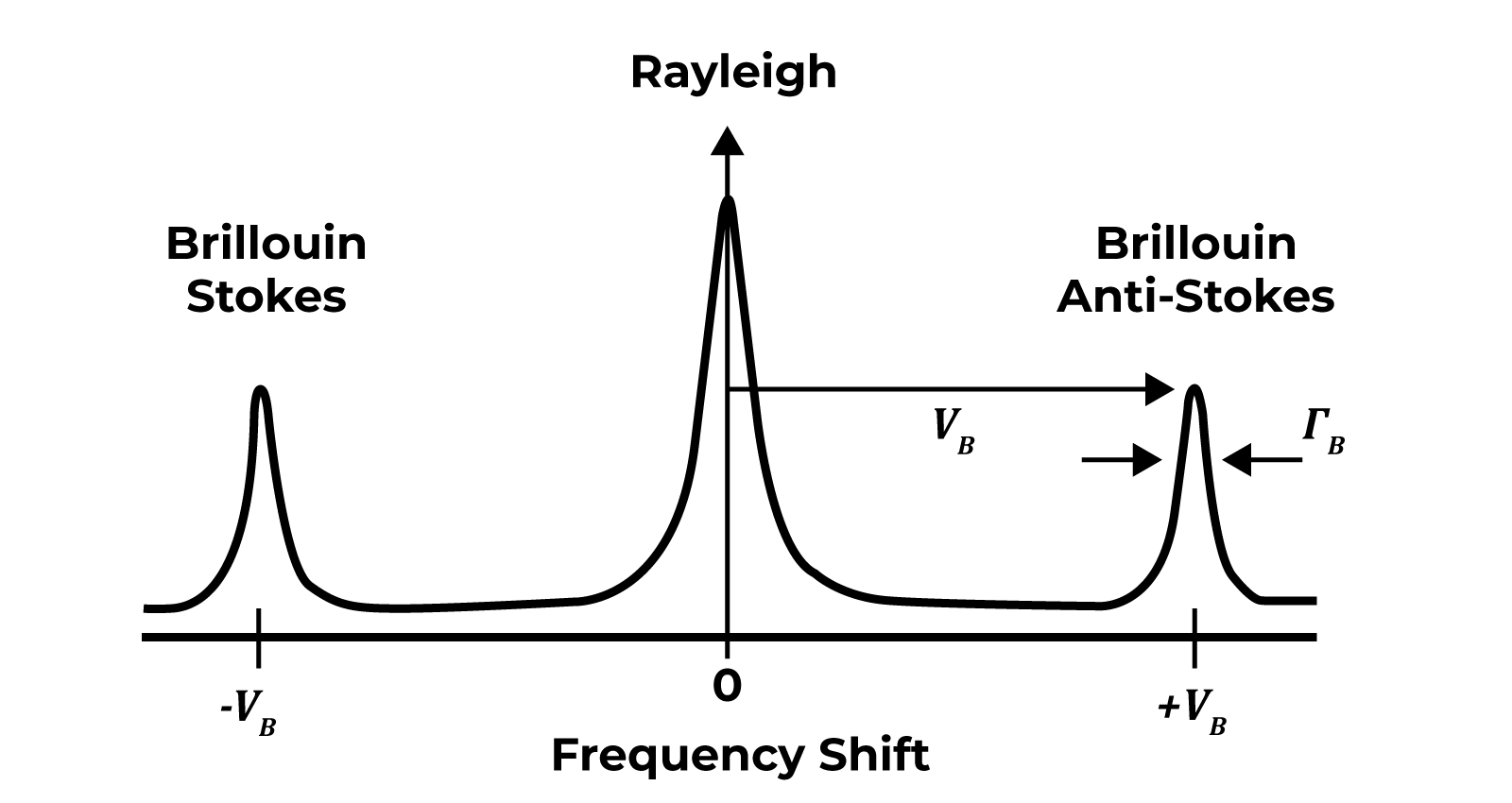
The Mechanical Signature
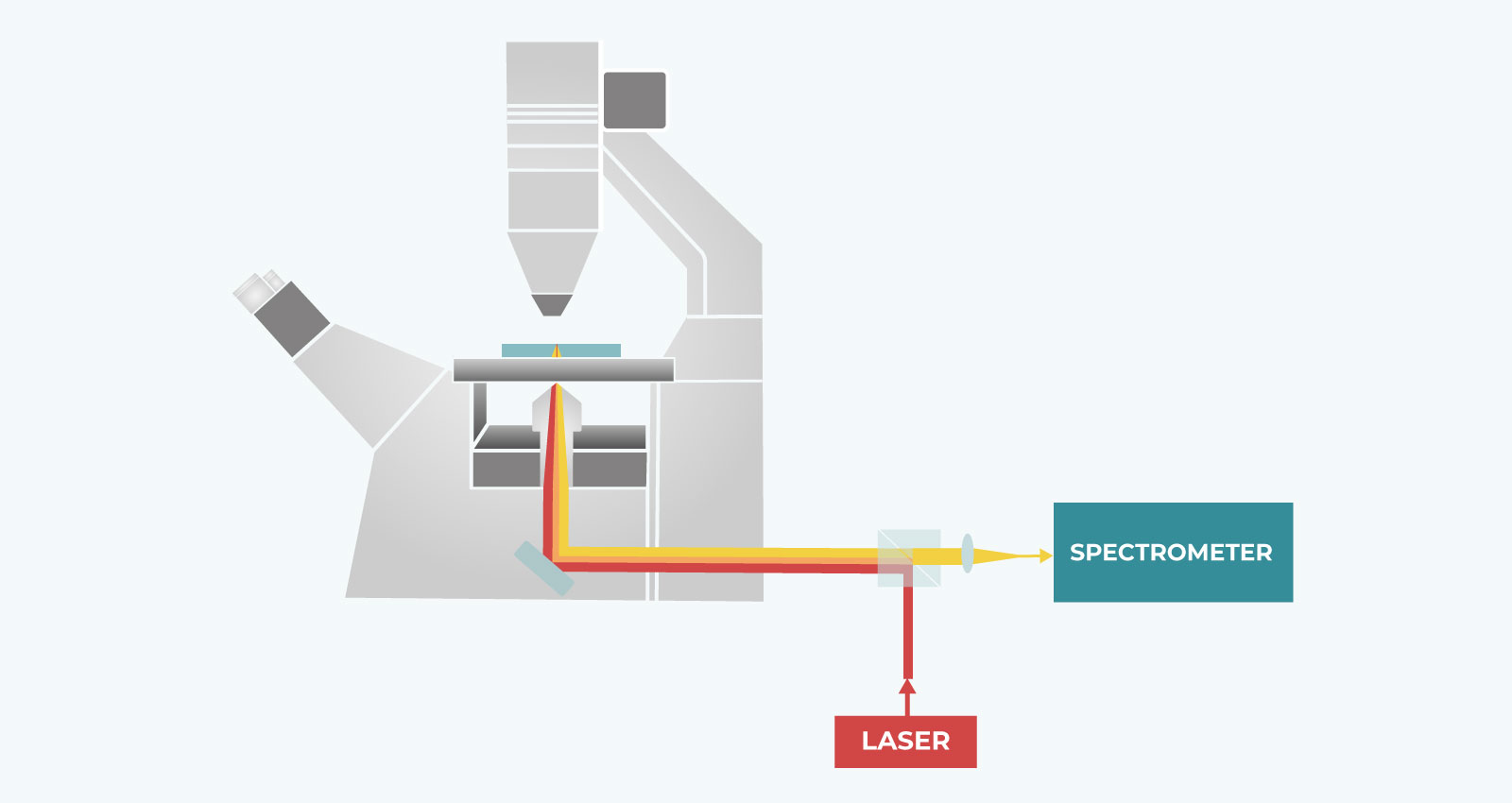
The Setup
A laser beam (red line) is focused on the sample, and the backscattered light (orange line) is collected by the same objective and analyzed by a high-resolution spectrometer. By scanning the laser beam on the sample plane, the complete characterization of the viscoelastic properties of the sample can be achieved through the analysis of its point-by-point Brillouin scattering spectrum.
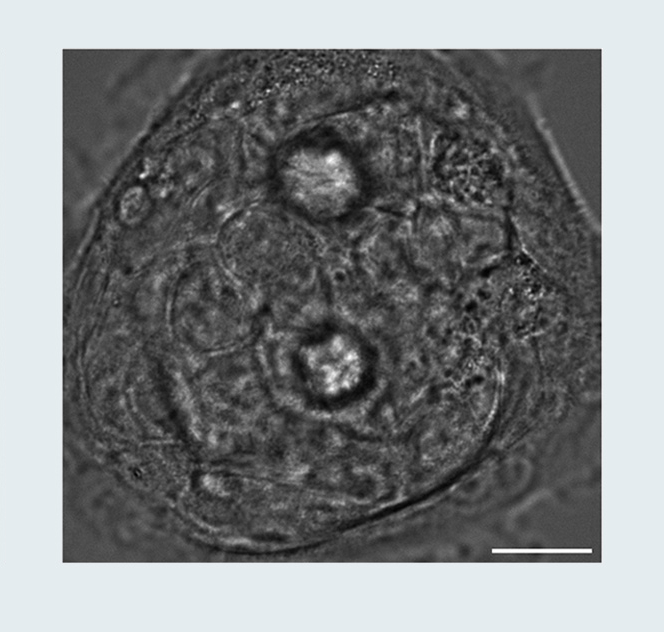
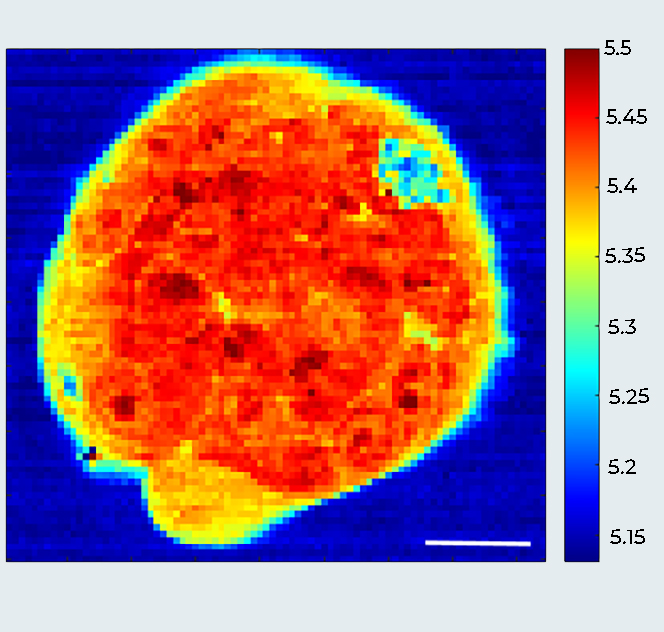
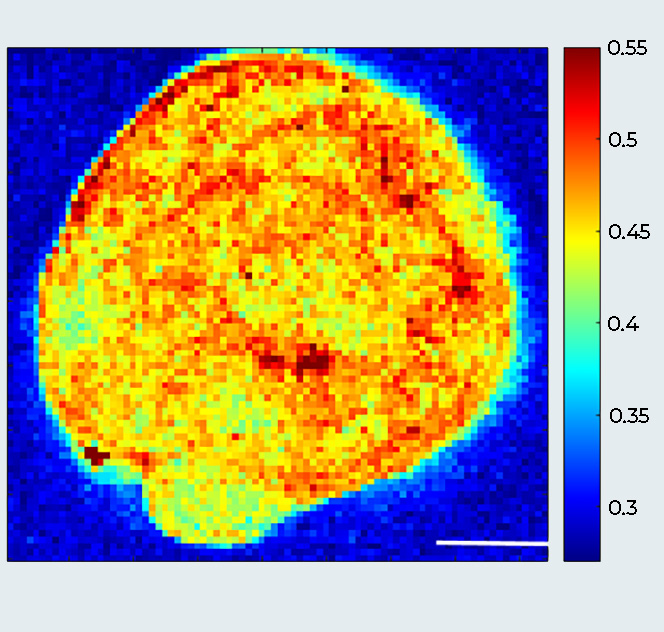
Breast Cancer Spheroid (MCF10DCIS.com cells): A Multimodal View
Sample courtesy: Prof. Giorgio Scita, IFOM Milano, Italy.
The Core Benefits of Brillouin Light Scattering in Life Sciences
- 100% Non-Contact: No cantilevers, no beads, no perturbation to your specimen.
- Label-Free: No exogenous dyes, preserving the physiological state of the specimen.
- Sub-Micron 3D Resolution: Penetrate deep into nuclei, organoids, and embryos to map the viscoelastic properties with sub-cellular resolution resolving power.
- Gentle on Live Samples: Uses 780 nm excitation to minimize phototoxicity, making it exceptionally well-suited for in-vivo Brillouin imaging.
- Versatile Integration: Compatible with standard microscope frames; enables fine Z-correlative imaging when combined with fluorescence spinning disk confocal technology.
Correlative Imaging: Brillouin + Spinning Disk Confocal
Structure Tells You WHO. Mechanics Tells You HOW.
By integrating Brillouin Spectroscopy with Confocal Spinning Disk Technology, you unlock a comprehensive view of your biological samples. This correlative approach bridges the gap between physical properties and structural organization. It empowers you to straightforwardly relate mechanical signatures, like stiffness and viscosity, to morphological features and cellular functions. Map this complex biological framework with absolute sub-micrometer precision in all three dimensions.
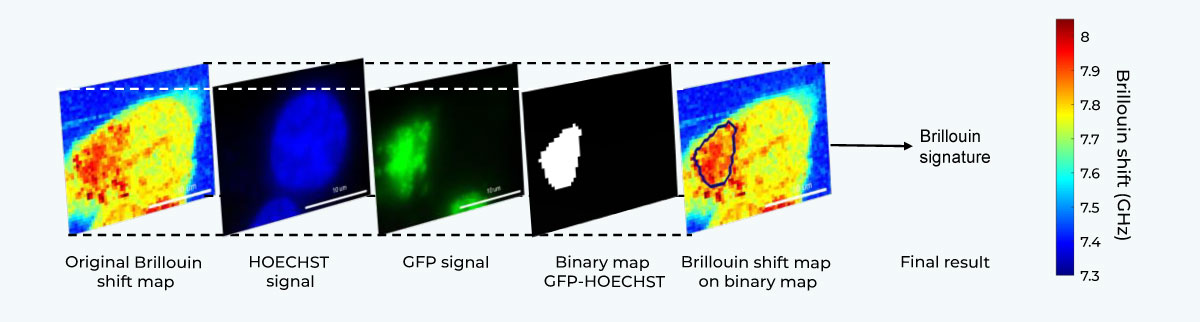
Correlative Imaging Workflow.
Brillouin Microscopy on living SK-N-BE cells expressing G3BP1 biomolecular condensates labeled with GFP. Confocal fluorescence maps (blue: nucleus, green: aggregates) acquired with CrestOptics X-Light V3 confocal spinning disk on Nikon Ti2. Brillouin shift maps. Scale bar 10um. Adapted from: Testi, C., Pontecorvo, E., Bartoli, C. et al. Stabilized real-time Brillouin microscopy reveals fractal organization of protein condensates in living cells. Nat Commun (2026). https://doi.org/10.1038/s41467-026-68984-2.
The combination of Brillouin Light Scattering with Spinning Disk Confocal Microscopy offers a promising direction for advancing our research in mechanobiology. This approach enables the simultaneous visualization of cellular structures alongside intravital measurements of their mechanical properties. While technically and conceptually demanding, such integration creates an opportunity to explore how forces and mechanical cues influence cell behavior and decision-making processes. Moving forward, a key challenge will be to refine these methods and to critically assess the extent to which they can resolve the complex interplay between mechanics and biological function.
— Prof. Paolo A. Netti and Carlo F. Natale (Department of Chemical, Materials and Industrial Production Engineering-DICMAPI, University of Naples Federico II, Naples, Italy)
Solving the Unsolvable
The Brillouin Insights
Since mechanical properties play a central role in biological processes, such as tumor progression, stem cell differentiation, and tissue morphogenesis, Brillouin microscopy is emerging as a powerful, non-invasive tool in different fields of modern mechanobiology and live-cell biophysics.
Below, we highlight key applications that benefit from Brillouin imaging.
Key Applications of Brillouin Light Scattering
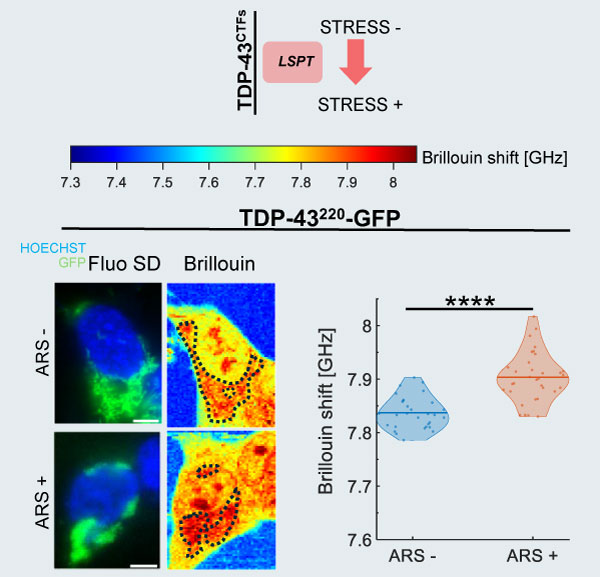
LLPS & LSPT in Neurodegenerative Diseases
LLPS & LSPT in Neurodegenerative Diseases
Brillouin microscopy enables study of Liquid-Liquid Phase Separation (LLPS) & Liquid-Solid Phase Transition (LSPT), allowing non-invasive characterisation of membrane-less organelles and their pathological transformations:
Identify the physical state of membrane-less organelles in their natural, in vivo state without external perturbation.
Distinguish between physiological liquid condensates and pathological aggregates based on their unique Brillouin signature in a label-free fashion.
Brillouin and confocal microscopy of living SK-N-BE cells.
SK-N-BE cells expressing different biomolecular condensates labeled with GFP. The panel highlights cells expressing TDP-43²²⁰ (TAR DNA-binding protein 43), which is known to be correlated to the formation of pathological aggregates in ALS (Amyotrophic Lateral Sclerosis) disease. The images combine confocal fluorescence maps (blue: nucleus, green: aggregates, acquired via X-Light V3 on a Nikon Ti2) with label-free Brillouin shift maps to reveal the local mechanical properties. Imaging parameters: 60x 1.42 NA objective. Scale bar: 5 µm. ARS = arsenite.
Adapted from: Testi, C., Pontecorvo, E., Bartoli, C. et al. Stabilized real-time Brillouin microscopy reveals fractal organization of protein condensates in living cells. Nat Commun (2026). https://doi.org/10.1038/s41467-026-68984-2
Nuclear Mechanobiology
Nuclear Mechanobiology
Brillouin imaging reveals how mechanical properties vary across nuclear compartments and influence cellular processes:
Map the longitudinal modulus of the nucleus in cellulo and quantify regional mechanical variations.
Evaluate the contribution of different nuclear compartments (nucleoli, nucleoplasm, heterochromatin foci) to overall nuclear mechanics.
Investigate how nuclear deformation impacts mechanical properties and cellular behaviour.
Understand the mechanical signatures underlying metastatic progression, migration, and cellular fate determination.
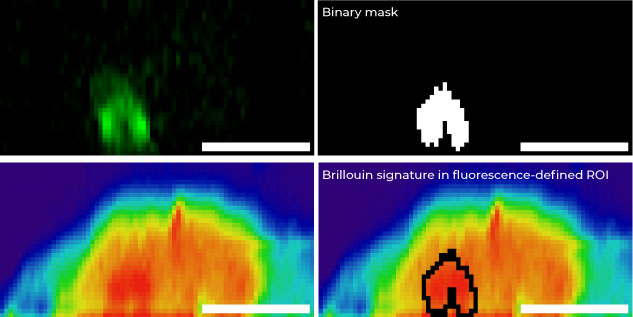
All-Optical Z-Sectioning of an Adipose Stem Cell.
Correlative 3D imaging of an Adipose Stem Cell (ASC). The panel combines a confocal fluorescence map (green: nucleolus, acquired via X-Light V3 on a Nikon Ti2) with a label-free Brillouin shift map.
As highlighted, the high-resolution fluorescence image can be seamlessly used to overlay a structural mask onto the Brillouin image, allowing for precise correlation between morphology and mechanical properties. Imaging parameters: 60x 1.42 NA objective. Scale bar: 5 µm. Sample courtesy: Dr. Carlo Natale, University of Naples Federico II, Italy.
Further Reading: for additional evidence on how Brillouin technology is successfully applied to study biomolecular condensates and nuclear mechanical stress, please refer to: Fasciani, A., D’Annunzio, S., Poli, V. et al. MLL4-associated condensates counterbalance Polycomb-mediated nuclear mechanical stress in Kabuki syndrome. Nat Genet 52, 1397–1411 (2020). https://doi.org/10.1038/s41588-020-00724-8
Tissue Alteration in Pathological Conditions
Tissue Alteration in Pathological Conditions
Brillouin microscopy provides a multi-scale view of tissue mechanics across healthy and diseased states:
Map viscoelastic properties throughout volumetric structures, including complex specimens such as 3D organoids and embryos.
Measure changes in tissue mechanics as indicators of disease onset and progression.
Investigate how mechanical stiffness influences developmental processes and tissue formation.
Identify mechanical gradients across tissue samples non-invasively, revealing structural heterogeneity.
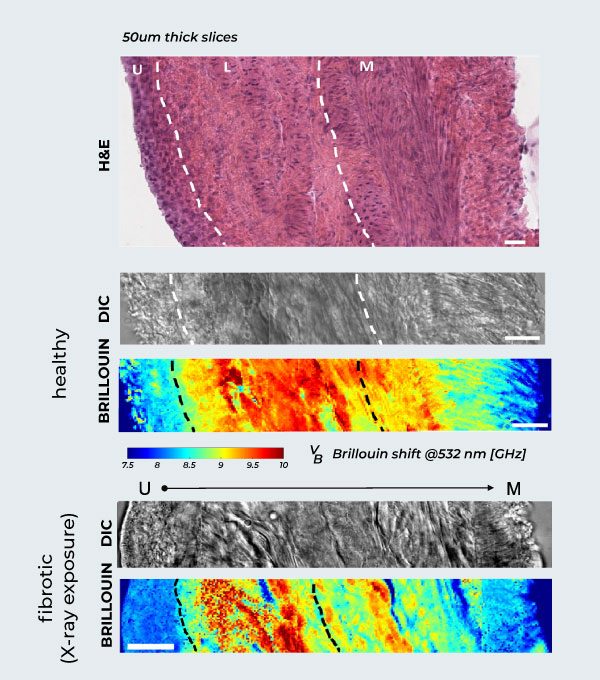
Brillouin Mapping of Radiation-Induced Actinic Cystitis.
Longitudinal evaluation of murine bladder walls following X-ray exposure. Healthy Tissue Anatomy: H&E (hematoxylin and eosin) staining of the healthy rat bladder wall, showing the different anatomical layers, alongside DIC and corresponding Brillouin maps from urothelium (left) to muscle (right). Biomechanical Progression: Correlative DIC and label-free Brillouin shift maps detailing fibrosis across the bladder wall layers (urothelium, lamina propria, muscle) at 2 months post-irradiation, compared to the healthy tissue above. Scale bars: 20 µm.
Adapted from: Martinez-Vidal, L., Testi, C., Pontecorvo, E. et al. Progressive alteration of murine bladder elasticity in actinic cystitis detected by Brillouin microscopy. Sci Rep 14, 484 (2024). https://doi.org/10.1038/s41598-023-51006-2
Scientific Publications Featuring Our Brillouin Technology
| Authors | Title | Journal | Year | Link |
|---|---|---|---|---|
| Alessandra Fasciani et al. | MLL4-associated condensates counterbalance Polycomb-mediated nuclear mechanical stress in Kabuki syndrome | Nature Genetics | 2020 | https://www.nature.com/articles/s41588-020-00724-8 |
| Laura Martinez-Vidal et al. | Progressive alteration of murine bladder elasticity in actinic cystitis detected by Brillouin microscopy | Scientific Reports | 2024 | https://www.nature.com/articles/s41598-023-51006-2 |
| Sarah D’Annunzio et al. | Chromatin condensates tune nuclear mechano-sensing in Kabuki Syndrome by constraining cGAS activation | BioRxiv | 2024 | https://www.biorxiv.org/content/10.1101/2024.05.06.592652v1.abstract |
| Léo Brechet et al. | On-the-fly adaptive SNR protocol to accelerate Brillouin microscopy | Optics Express | 2025 | https://opg.optica.org/oe/fulltext.cfm?uri=oe-33-13-28511 |
| Pierre Bouvet et al. | Consensus statement on Brillouin light scattering microscopy of biological materials | Nature Photonics | 2025 | https://www.nature.com/articles/s41566-025-01681-6 |
| Noemi Svolacchia et al. | Cell wall–derived mechanical signals control cell growth and division during root development | Science Advances | 2025 | https://www.science.org/doi/full/10.1126/sciadv.aea8647 |
| Chiara Bartoli et al. | Real-Time Brillouin Microscopy for Linewidth Imaging of Protein Condensates in Living Cells | IEEE Photonics Journal | 2026 | https://ieeexplore.ieee.org/abstract/document/11358959 |
| Claudia Testi et al. | Stabilized real-time Brillouin microscopy reveals fractal organization of protein condensates in living cells | Nature Communications | 2026 | https://www.nature.com/articles/s41467-026-68984-2 |
We, as CrestOptics, are committed to being part of the Brillouin community.
For more detailed information, see the original Consensus paper:
Bouvet, P., Bevilacqua, C., Ambekar, Y. et al. Consensus statement on Brillouin light scattering microscopy of biological materials. Nat. Photon. 19, 681–691 (2025). https://doi.org/10.1038/s41566-025-01681-6.
Technical Authority
Comparison Table: Brillouin Microscopy vs AFM nano indentation vs Optical Tweezers
| Technique | Contact Y/N (Forces applied) | Depth in biological sample | Max lateral (XY) resolution | Max axial (Z) resolution | Time to map 10×10 um2* |
|---|---|---|---|---|---|
| Brillouin Microscopy | No (optical) | ≈ 150 um (clear), 40-80 um (tissue) | 300 nm | ≈ 1.0 um | ≈ 1 min |
| AFM nano indentation | Yes (uN – nN) | Surface only | 20-30 nm | ≤ 100 nm indentation | ≈ 3-5 h |
| Optical Tweezers | Yes, Bead-mediated (0.1-200 pN) | ≤ 50-100 um (bead insertion) | 3-10 nm (tracking) | 10-20 nm (tracking) | ≈ 30 s |
*Assumes raster or holographic scan at native lateral step over a 10 um × 10 um field. Brillouin: confocal, 100 ms/voxel. AFM: 1s per indentation on 100 nm grid (≈3-5 h). Optical tweezers: holographic scan with 1kHz bead tracking (∼10-30 s). Depth values: Zhong 2019; Favre-Bulle 2017. Other parameters from Kabakova 2024, Zanini 2024, and standard specs.
Synergy, Not Substitution: The Power of Correlative Mechanics
Every technique has its unique advantages and specific use cases. Brillouin imaging is not designed to replace traditional mechanobiology tools like AFM or Optical Tweezers, but rather to elevate them. While other methods might excel at surface-level contact measurements, Brillouin provides non-destructive, 3D internal mapping. By embracing a correlative approach, you can synergize data across different platforms, leveraging the strengths of each to build a complete, multimodal understanding of your sample.
You ask, We answer.
☑ What is the spatial resolution of Brillouin imaging? — Sub-micron, diffraction-limited.
☑ Can Brillouin be used on live cells? — Yes, it is designed for real-time in vivo monitoring.
☑ How does it complement Spinning Disk? — By providing mechanical context to structural data.
Early Access Program (EAP)
Secure Your Place at the Cutting Edge of Mechanobiology
CrestOptics is opening a restricted invitation for Key Opinion Leaders and leading research institutions to integrate our premium Brillouin technology ahead of the global commercial launch.
This program is designed for laboratories that require an immediate strategic advantage in 3D biomechanical mapping.
Shape the Future of Mechanobiology with us obtaining the following privileges:
- Strategic Partnership: Secure the technology ahead of global launch with a dedicated collaboration pricepoint.
- Direct R&D Channel: Continuous technical support and feedback loop with our lead optical and software engineers.
- Data Validation Support: Expert review of your datasets to optimize acquisition parameters for specific biological samples.
- Co-Publication Opportunities: Support for early-stage application notes and technical contributions to your research papers.