At CrestOptics, we are deeply committed to staying at the forefront of scientific progress, dedicating our utmost effort to innovation. Our mission is to develop cutting-edge technologies that are both reliable and adaptable to the ever-evolving needs of researchers. The market itself recognizes our drive for innovation: in the past three years, we have introduced new solutions in spinning disk technology and super-resolution microscopy, expanding imaging capabilities for accessibility and resolution performance. Building on this foundation, we continue to explore and integrate into emerging imaging technologies that open new frontiers in biomedical and biophysical research.
Holotomography is a smart example of these emerging technologies, offering a powerful approach to a non-invasive live-cell imaging. This technology enables researchers to visualize cellular structures in three dimensions with exceptional clarity. By providing quantitative phase information, holotomography captures structural details that would otherwise be difficult to observe, offering a deeper understanding of cellular morphology and dynamics.
At CrestOptics, we are deeply committed to staying at the forefront of scientific progress, dedicating our utmost effort to innovation. Our mission is to develop cutting-edge technologies that are both reliable and adaptable to the ever-evolving needs of researchers. The market itself recognizes our drive for innovation: in the past three years, we have introduced new solutions in spinning disk technology and super-resolution microscopy, expanding imaging capabilities for accessibility and resolution performance. Building on this foundation, we continue to explore and integrate into emerging imaging technologies that open new frontiers in biomedical and biophysical research.
Holotomography is a smart example of these emerging technologies, offering a powerful approach to a non-invasive live-cell imaging. This technology enables researchers to visualize cellular structures in three dimensions with exceptional clarity. By providing quantitative phase information, holotomography captures structural details that would otherwise be difficult to observe, offering a deeper understanding of cellular morphology and dynamics.
With its ability to deliver high-resolution 3D imaging in real time, holotomography is revolutionizing the way researchers study live cells. It allows scientists to analyze cellular behavior over extended periods without the risk of phototoxicity. This makes it particularly valuable for applications that require long-term imaging, such as cell differentiation studies, drug response monitoring, and the investigation of dynamic intracellular processes.
Until now, fluorescence imaging integrated with holotomography has been limited to Widefield (WF) fluorescence, restricting its applications. WF fluorescence lacks the necessary resolution in the Z-axis to achieve true structural-functional colocalization. Additionally, its high phototoxicity and rapid photobleaching create limitations for long-term live-cell studies. To overcome these limitations, we have partnered with Tomocube, Inc., a global leader in holotomography imaging, to push the boundaries of correlative microscopy.
To further enhance imaging capabilities, holotomography is also combined with fluorescence microscopy: this combination allows researchers to correlate quantitative, labelfree imaging with precise molecular-functional information, unlocking a more complete understanding of biological processes.
3D reconstruction of HT and SDCM images. This image was acquired with a 40x air objective 0.95 NA.
3D reconstruction of HT and SDCM images. This image was acquired with a 40x air objective 0.95 NA.
We are proud to present the first-ever correlative Spinning-Holotomography system, combining the CrestOptics X-Light V2 Spinning Disk Confocal Microscopy (SDCM) and Tomocube’s HT-X1™ Plus Holotomography (HT) system. This innovative approach delivers high-speed, high-resolution 3D imaging while minimizing phototoxicity, providing researchers with a powerful, holistic tool to capture molecular and structural information from a whole sample.
Fill the form to get the full Application Note
As the first in the 2nd-generation Holotomography series, the HT-X1™ revolutionizes biomedical research with high-resolution 3D imaging and exceptional stability. Its versatile platform supports various multi-well plates and offers an extended axial scan range up to 150 microns —surpassing the 40-micron limit of the first-generation HT. Powered by TomoAnalysis™ software, the HT-X1 equips researchers with advanced tools for quantitative analysis and reliable imaging.
Now, the HT-X1™ Plus advances this technology further, delivering clearer, more detailed imaging with enhanced optics, improved reconstruction algorithms, and additional user-friendly features. With a high-spec camera offering a 4x larger field of view and faster acquisition, it excels in high-throughput phenotypic screening using multi-well plates. The HT-X1™ Plus extends holotomography to dense organoids, tissue sections, and fast-moving microorganisms, empowering researchers with the precision, efficiency, and reliability to drive biomedical discovery.
Based on market feedback regarding the limitations of WF and the growing need for a gentle 3D fluorescence solution capable of covering the new scale of macro samples generated by holotomography, CrestOptics adapted the X-Light V2 to seamlessly integrate within Tomocube’s hardware and software. The system was designed to provide multi-color fluorescence imaging with up to seven excitation wavelengths while being equipped with high-sensitivity cameras to ensure optimal signal detection. Spinning disk confocal fluorescence overcomes the WF fluorescence limitations by offering Z-resolution comparable to holotomography, allowing researchers to achieve precise colocalization of molecular and structural details across the entire sample volume. At the same time, its gentle illumination minimizes phototoxicity and photobleaching, making it ideal for extended live-cell imaging.
Building on these advantages, this Application Note will showcase two distinct applications that leverage the combined strengths of SDCM and HT. By bridging the gap between fluorescence and quantitative phase imaging, this approach unlocks new possibilities for live-cell analysis, dynamic interaction studies, and volumetric imaging of complex biological samples.
1. Cell monolayer imaging and subcellular organelles interaction analysis
In this first application, we demonstrate how combining SDCM and HT maximizes the amount of data that can be extracted from HeLa cell analysis, optimizing the efficiency and depth of the study.
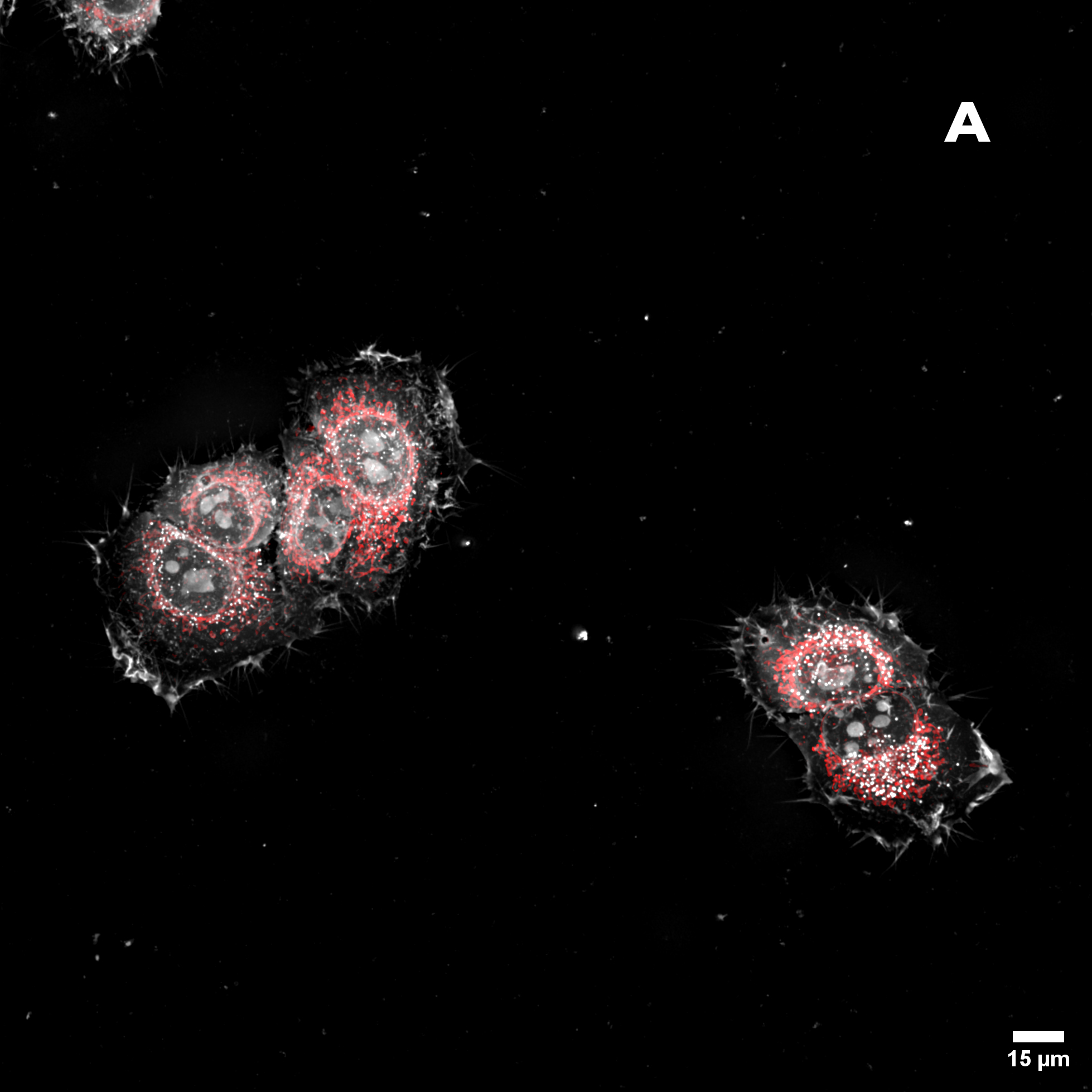
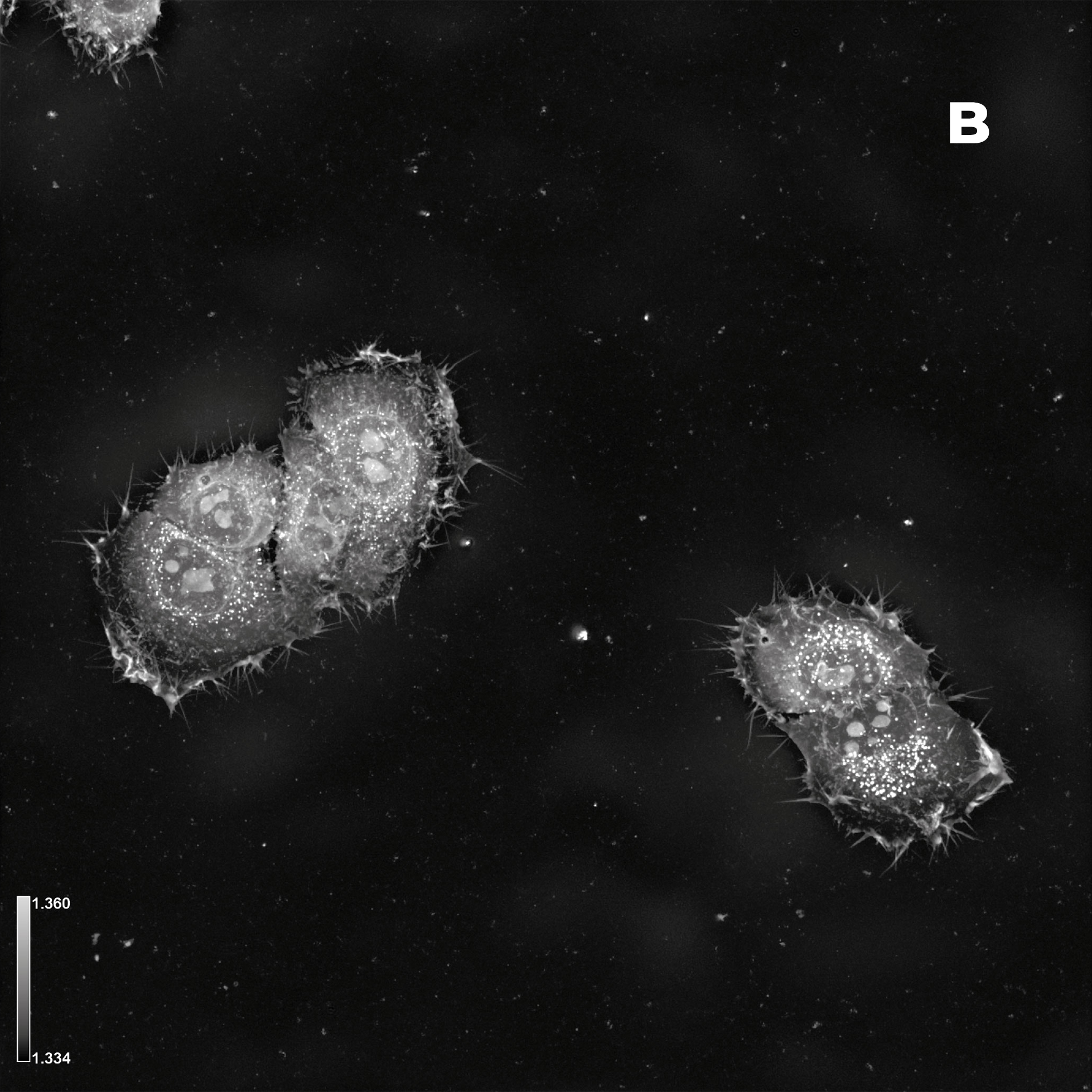
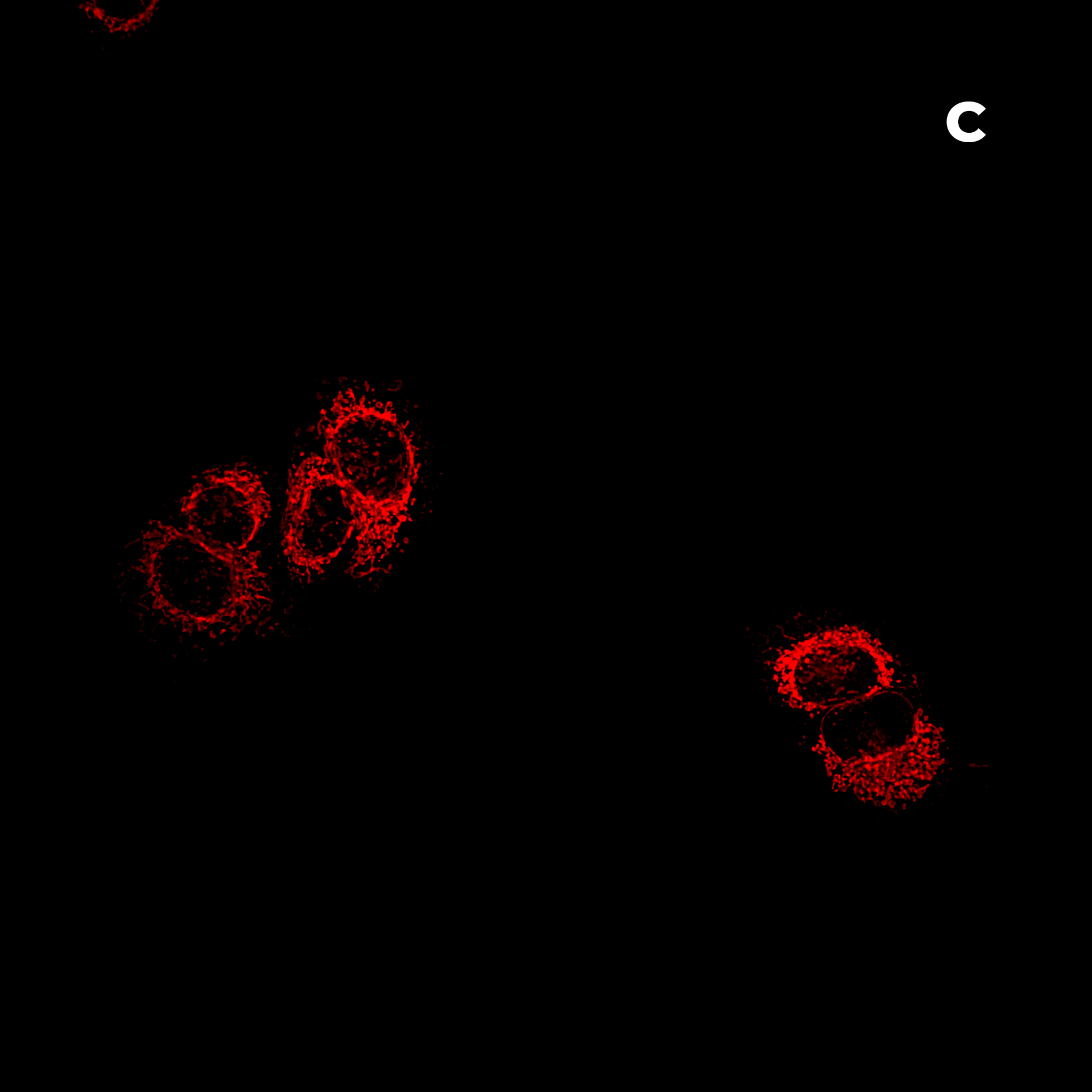
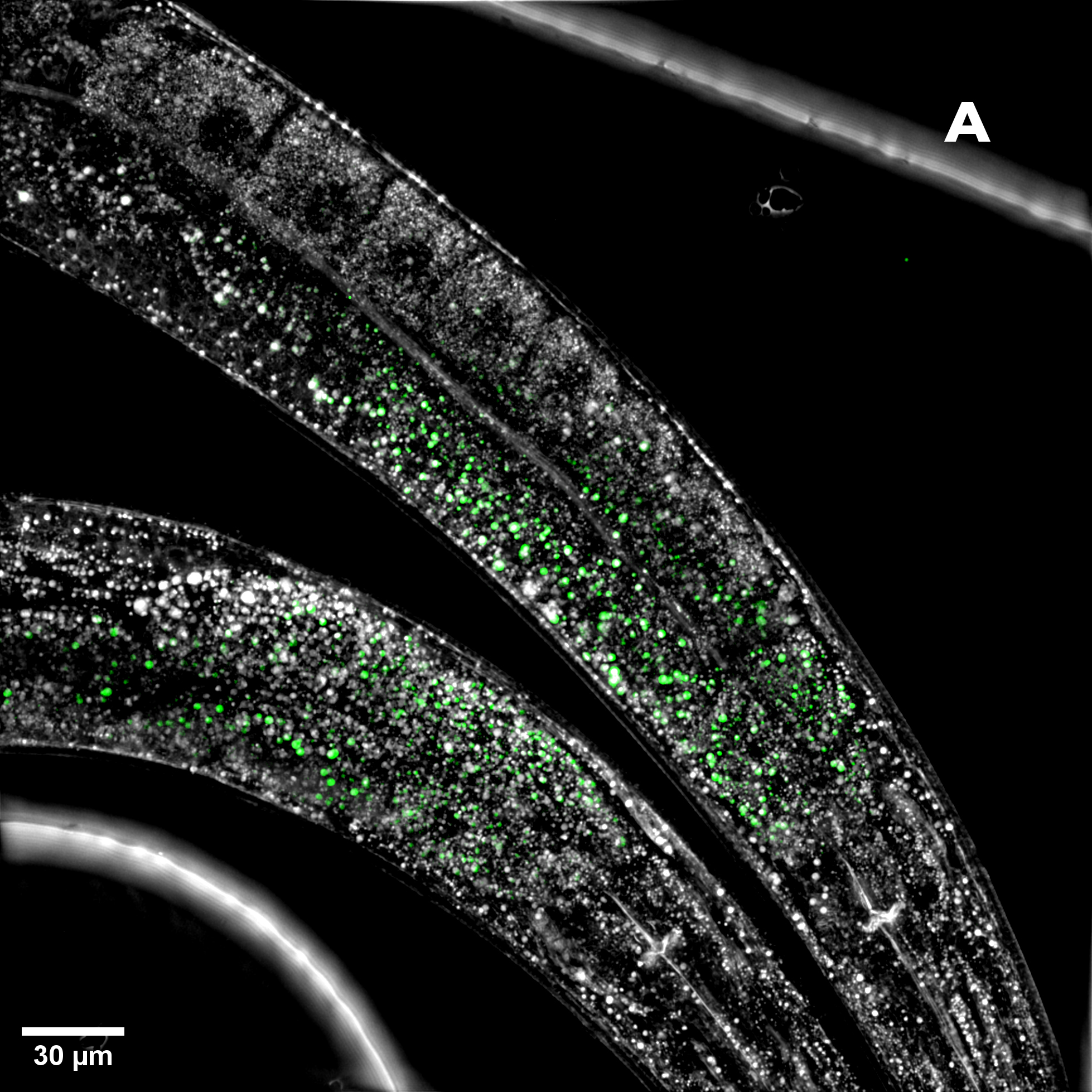
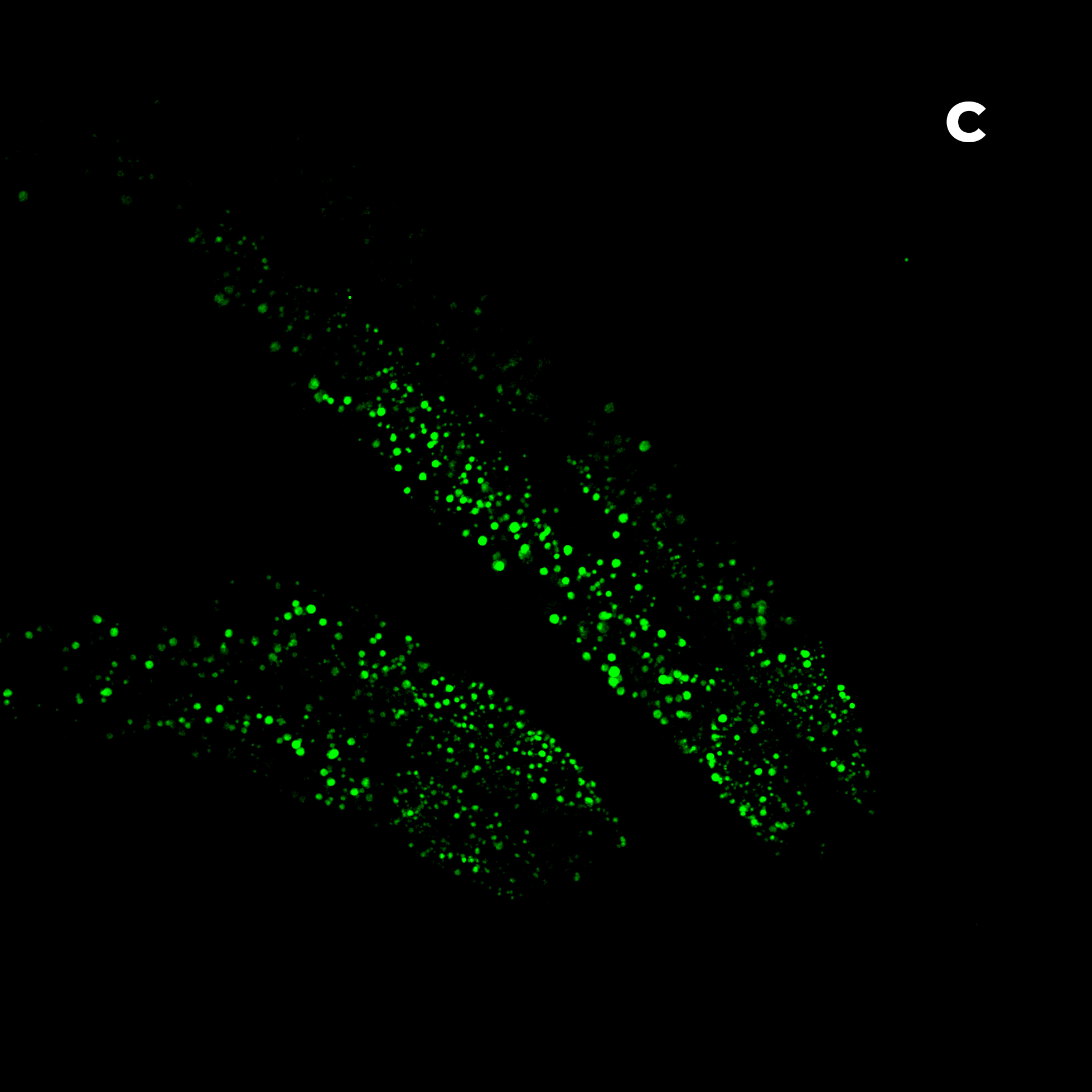
Figure 2: Hela cells monolayer. A) HT (gray) + SDCM (light blue) MIP. B) HT image. C) SDCM image of mitochondria (light blue). This image was acquired with a 40x air objective 0.95 NA.
Figure 2A presents a Maximum Intensity Projection (MIP) of HeLa cells acquired using both HT and SDCM. In Figure 2B, the HT image reveals the architecture of subcellular structures—such as nuclei, mitochondria, and lipid droplets, thanks to the high resolution of the HT-X1™ Plus. Figure 2C shows mitochondria staining imaged with SDCM, where the optical sectioning capability of the X-Light V2 enables precise identification and localization of mitochondria within the cell. This example highlights the potential of combining these two techniques to enhance correlative imaging and provide a more comprehensive view of cellular structures.
The high lateral resolution of HT, combined with the high axial resolution of SDCM, enables the detailed study of interactions between subcellular components. Organelle interactions play a crucial role in various cellular functions, including the adaptive response to nutrient availability. In particular, mitochondria and lipid droplets engage in dynamic, highly regulated interactions that respond to changes in the cellular metabolic state [2]. These dynamic interactions play a key role in processes such as energy storage, oxidative stress response, and adaptation to nutrient availability, making them highly relevant in research on obesity, diabetes, and neurodegenerative diseases [3]. The combination of HT and SDCM enables clear identification of both mitochondria (detected via fluorescent staining) and lipid droplets, which appear in HT as small, round, white structures. HT is particularly well suited for studying lipid droplets, as it distinguishes cellular components based on their refractive index. Lipid droplets, having a relatively high refractive index, appear as bright spherical structures.
By leveraging the strengths of both techniques, HT and SDCM enable a detailed investigation of mitochondria-lipid droplet interactions using just a single fluorescent marker. Additionally, this combination facilitates long-term live imaging, allowing the study of these interactions over time with minimal perturbation to the sample.
2. Whole model organism volumetric imaging
In this second application, we demonstrate how the combination of the two instruments allows the acquisition of three-dimensional images of thick samples.
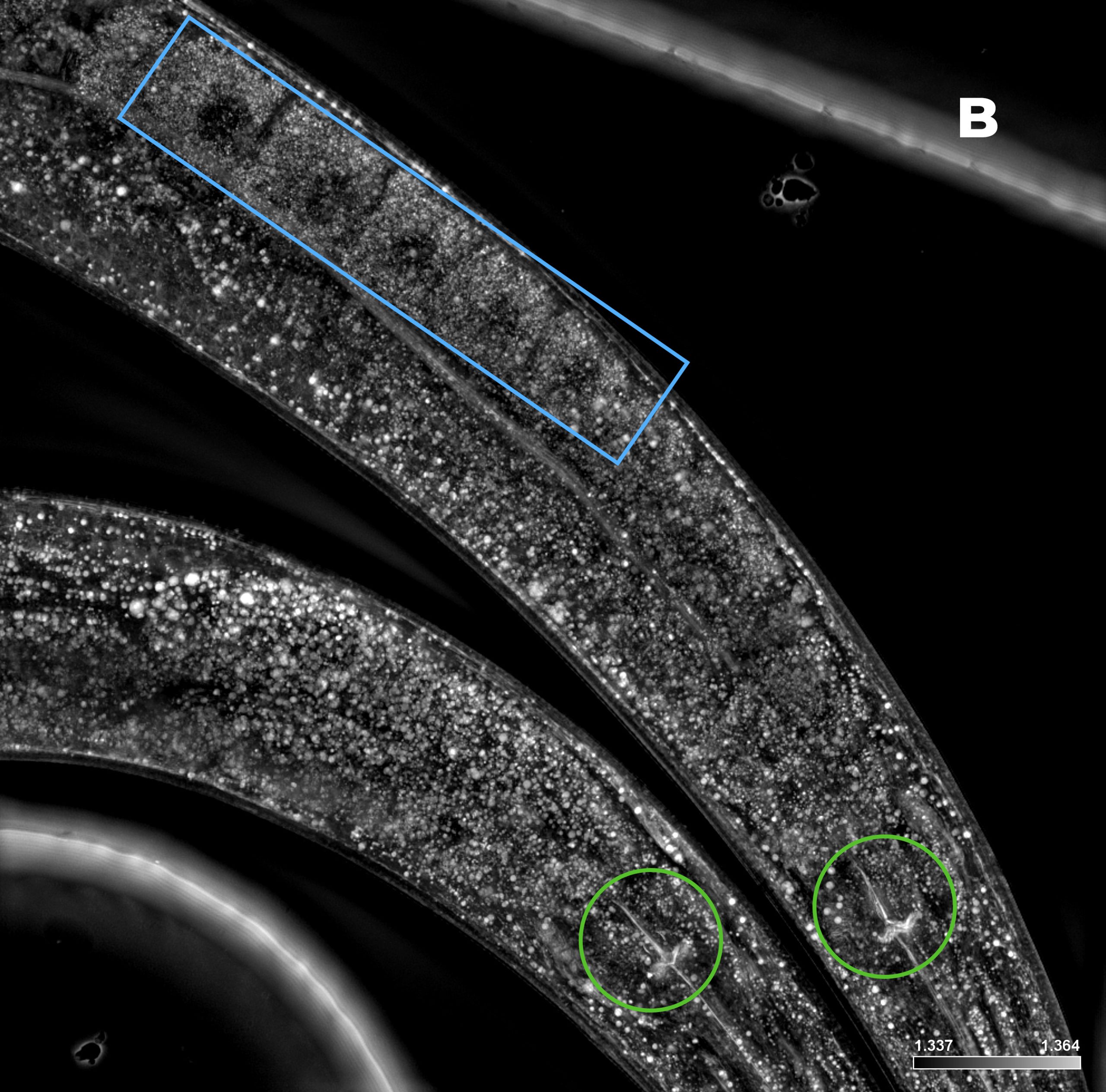
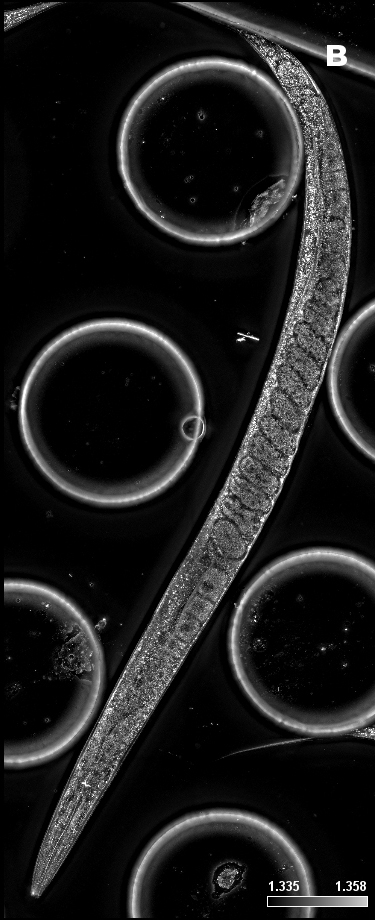
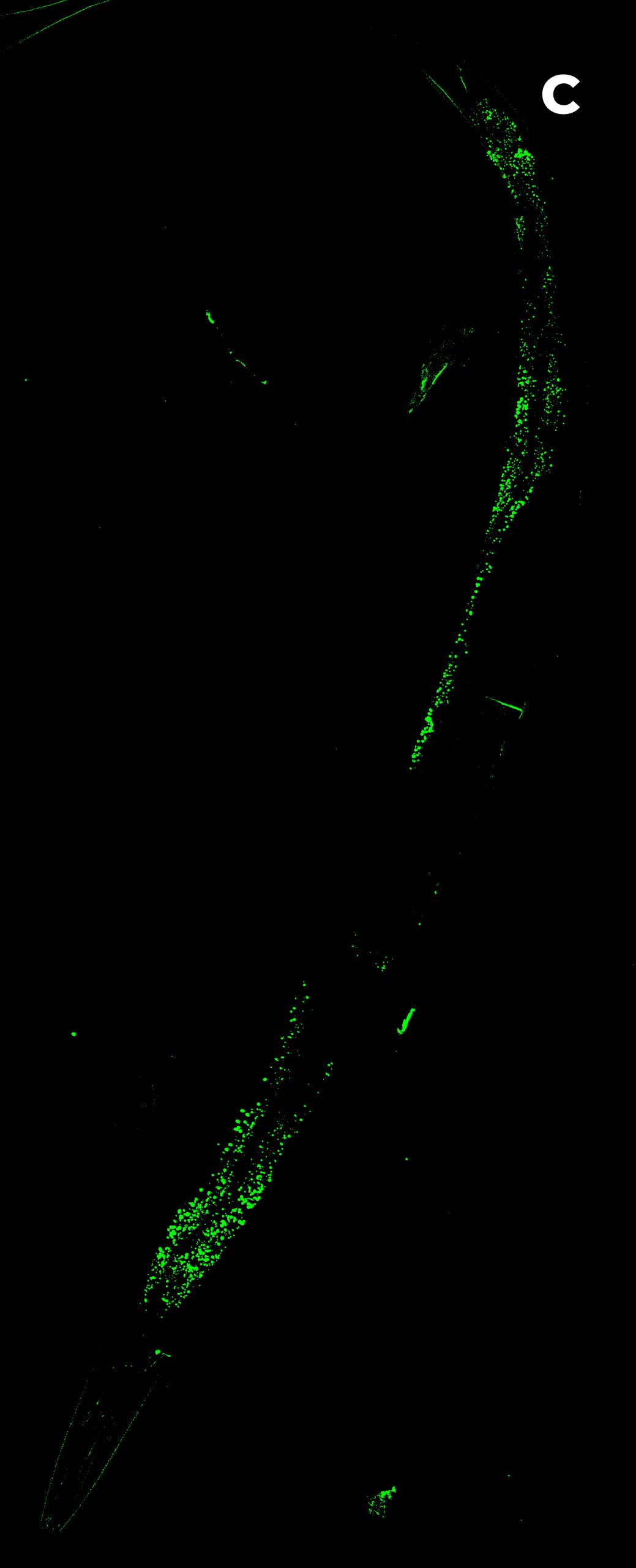
Figure 3: C. elegans. A) HT (gray) + SDCM (green) MIP of 150 μm. B) HT image. Blue rectangle: eggs; green circles: pharynx. C) SDCM image of bacteria (green). D) 3D reconstruction of HT and SDCM mages. This image was acquired with a 40x air objective 0.95 NA.
Figure 3A shows the MIP of a z-stack acquired on a live sample of C. elegans with both the HT and the SDCM. In Figure 3B, the HT image provides a highly detailed view of the anatomical structures of C. elegans, allowing clear identification of key components such as the pharynx (highlighted by green circles) and the eggs (blue rectangle). In Figure 3C, bacteria inside C. elegans, acquired via SDCM, are distinctly visible. By merging the two images, these bacteria can be precisely localized in relation to the anatomical structures of the organism, offering valuable spatial context for biological analysis.
The advanced optical sectioning capabilities of both the X-Light V2 and HT-X1™ Plus enable high-speed 3D imaging and in-depth structural analysis of thick specimens, such as the one presented here. This capability is further demonstrated in Figure 3D, which shows a 3D reconstruction of C. elegans obtained through the combined use of HT and SDCM. By taking advantage of the strengths of both techniques, this approach facilitates a comprehensive and high-resolution investigation of complex biological samples.
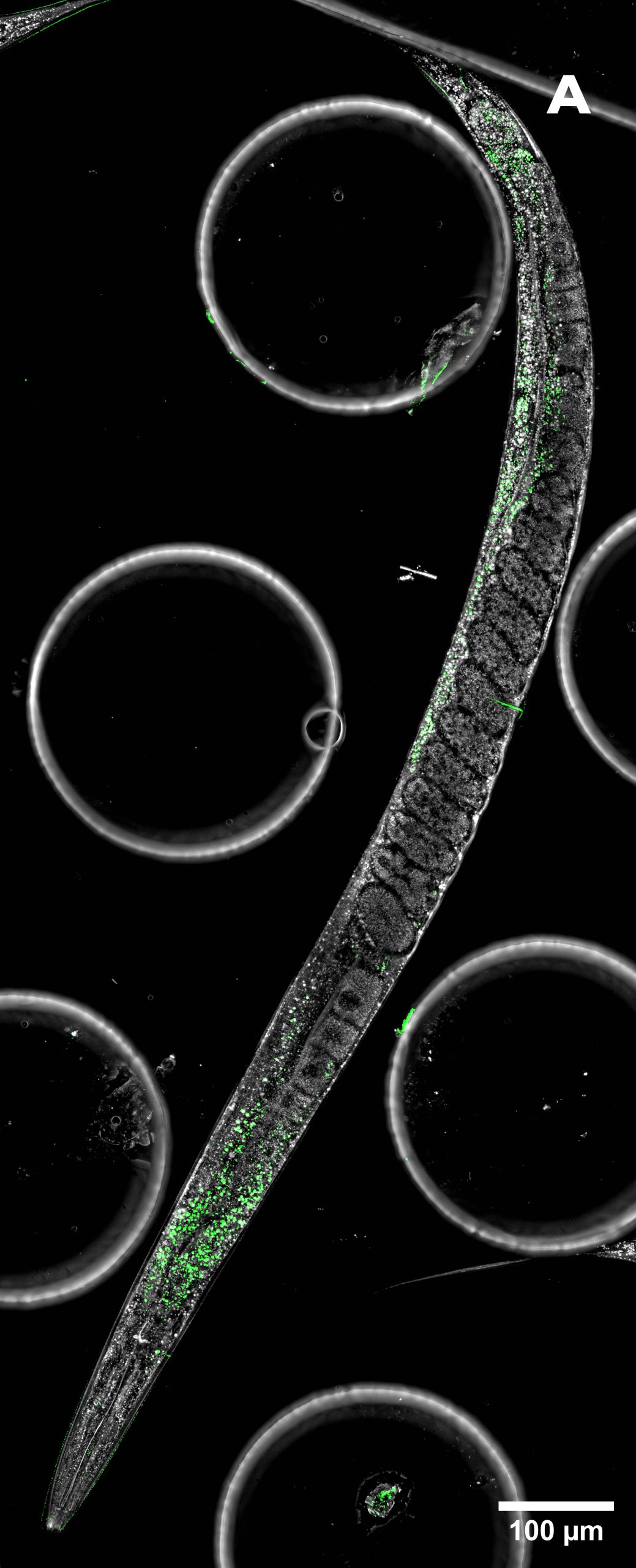
The combination of 3D imaging and stitching, utilizing both HT and SDCM, significantly enhances the amount of information that can be extracted from a single sample. As shown in Figure 4, the use of stitching allowed the reconstruction of an entire C. elegans specimen, enabling a comprehensive analysis of its full anatomical structure. This approach not only provides a broader spatial context but also preserves high-resolution details across large fields of view, making it a powerful tool for studying complex biological systems at multiple scales.
Figure 4: large image of C. elegans. A) HT (gray) + SDCM (green) MIP of 120 μm. B) HT image. C) SDCM image of bacteria (green). D) 3D reconstruction of HT and SDCM mages. This image was acquired with a 40x air objective 0.95 NA.
Conclusion
The integration of holotomography HT and spinning disk confocal microscopy SDCM represents a significant advancement in high-resolution, correlative imaging for biomedical research. By combining the molecular specificity of fluorescence imaging with the label-free, high-contrast structural information provided by holotomography, researchers can obtain a more comprehensive understanding of cellular and tissue architecture. This approach enables the acquisition of morphological and molecular data, enhancing studies on subcellular interactions and whole-organism imaging. The ability to perform high-speed 3D imaging, live-cell analysis and large-scale reconstructions with a single setup makes this combination a powerful and versatile tool for a wide range of applications. As demonstrated in this Application Note, the synergy between the X-Light V2 and the HT-X1™ Plus expands the frontiers of quantitative imaging, providing researchers with unprecedented insights into complex biological systems.
Acknowledgments
The C. elegans samples where kindly provided by: Viola Folli, Enrico Lanza and Valeria Lucente.
This Application Note is the result of a collaborative effort between CrestOptics and Tomocube, Inc. We sincerely thank Bruno Combettes and Daniel Ghete for their invaluable support in both generating the data and contributing to the writing of this work.
Bibliography
[1] Kim, G., Hugonnet, H., Kim, K. et al. Holotomography. Nat Rev Methods Primers 4, 51 (2024). doi.org: 10.1038/s43586-024-00327-1
[2] Song JE, Alves TC, Stutz B, Šestan-Peša M, Kilian N, Jin S, Diano S, Kibbey RG, Horvath TL. Mitochondrial Fission Governed by Drp1 Regulates Exogenous Fatty Acid Usage and Storage in Hela Cells. Metabolites. 2021 May 18;11(5):322. doi: 10.3390/metabo11050322. PMID: 34069800; PMCID: PMC8157282.
[3] Fan H, Tan Y. Lipid Droplet-Mitochondria Contacts in Health and Disease. Int J Mol Sci. 2024 Jun 22;25(13):6878. doi: 10.3390/ijms25136878. PMID: 38999988; PMCID: PMC11240910.