The transition from 2D to 3D cell cultures marked a significant breakthrough in biological and medical research. Traditional 2D cultures, where cells grow on flat surfaces, have been instrumental in studying cell behaviour, but they fail in mimicking the complex, three-dimensional environment of living tissues.
3D cultures indeed, provide a more accurate representation of in vivo conditions, offering numerous advantages over 2D systems. One of the main benefits of 3D cell cultures is their ability to better simulate the physiological conditions of tissues and organs, leading to more relevant biological insights. Cells in 3D cultures exhibit more realistic morphology, gene expression, and cellular interactions. This makes 3D systems superior for studying cancer, drug development, and tissue engineering, where cellular behaviour plays a critical role.
Neural rosettes are radial arrangements that mimic the organization of the neural tube during early embryonic development. They are usually cultured from pluripotent stem cells, such as induced pluripotent stem cells (iPSCs). The process begins by differentiating these stem cells into neural progenitor cells (NPCs) through the use of specific growth factors and signalling molecules, which simulate the cues present during early neural development. These structures represent a critical stage in the development of the central nervous system, where neural stem cells begin to differentiate into neurons and other specialized cell types.
The development of neural rosettes is characterized by NPC apical-basal polarization and the formation of a lumen. It closely resembles the neural tube’s central canal during early stages of embryonic brain and spinal cord development. In this context, the lumen represents the future ventricular system of the brain, where cerebrospinal fluid will eventually flow. The NPC polarization and collective formation of a lumen is a key step in neurodevelopment, and it serves as a critical marker of correct structural formation, providing insights into early neural patterning and development.
Neural rosettes are a hallmark of neurogenesis and are often used to initiate cultures for studying processes such as neuronal differentiation, migration, and the formation of neural networks. By using spheroids initially comprised of neural rosettes, scientists can gain deeper insights into brain development, disease modelling, and the potential effects of drugs or genetic mutations on neural tissues.
In this context, the need arises to create tools for the rapid screening of neural rosettes in order to rapidly study their development and to evaluate the effect on them of particular drugs.
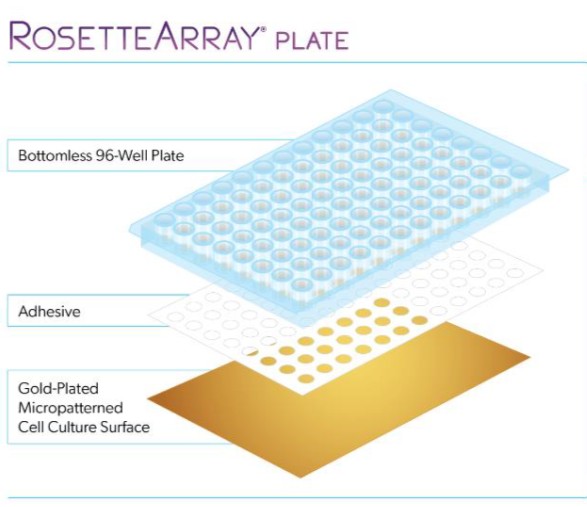
Figure 1. Left: Schematic showing the structure of micropatterned multiwells for neural rosettes screening. Right: image of a RosetteArray plate
Neurosetta has commercialized this platform to screen the early stages of human brain and spinal cord development in a reproducible, cost-effective and high-throughput manner.
The aim of this Application Note is to demonstrate that RosetteArray® plates, combined with the X-Light V3, represent a powerful and efficient tool for fast and accurate screening of neural rosettes.
As shown in Figure 2, the large field of view (FOV) and stitching capabilities of the X-Light V3 allowed us to image all the neural rosettes within a single well. This type of acquisition provides a rapid, preliminary screening of the rosettes across different wells, enabling us to quickly verify how many rosettes have successfully formed. Furthermore, as highlighted in the circle in Figure 2, we can assess the number of rosettes in which a lumen has developed. This information is crucial for determining the developmental stage of each rosette. After gathering this initial data, we can then focus on the rosettes with a fully formed lumen, increasing magnification to enhance the level of detail for more in-depth analysis.

Figure 2. Large image of neural rosettes stained for Phalloidin. This image was acquired with CFI Plan Apochromat Lambda D 20x air objective (0.8 NA, 0.8 WD). In the circle it is shown an enlargement of a neural rosette in which the lumen was formed
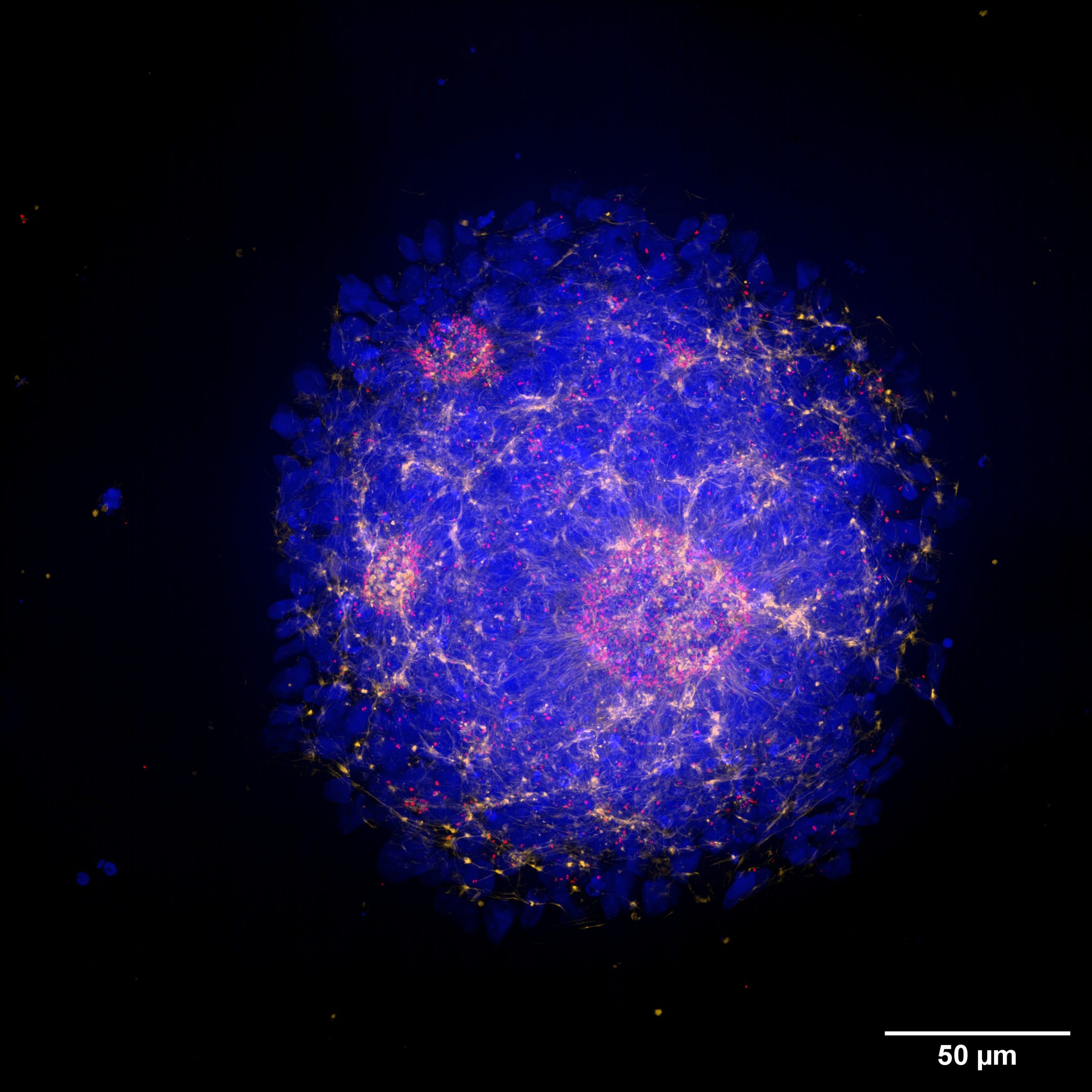
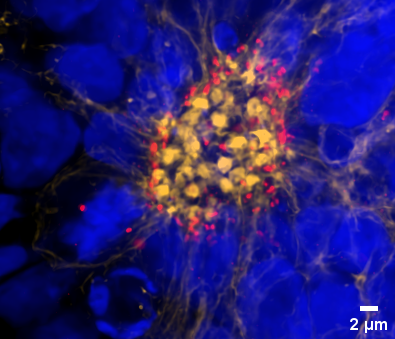
The 25 mm diagonal FOV of the X-Light V3, allowed us to go into more detail and image an entire neural rosette with a single image at 60x magnification. Figure 3 shows the Maximum Intensity Projection (MIP) of a neural rosette with a staining for phalloidin (yellow) and pericentrin (red).
Figure 3. MIP (40 µm) of a neural rosette imaged with a Nikon CFI Plan Apochromat Lambda D 60X oil objective (1.42 NA, 0.15 WD). Neural rosettes are stained with: DAPI (blue), Phalloidin (yellow) and Pericentrin (red)
By using markers for pericentrin, a protein that localizes to the centrosome and recruits other proteins to ensure the proper formation of the centrosome and mitotic spindle, further insights can be gained into cell maturation within the rosette. Specifically, the orientation of the mitotic spindle influences the cleavage plane and predicts cell fate decisions, determining whether the divisions will be symmetric and proliferative or asymmetric and differentiative. This information is crucial for determining the developmental status of a neural rosette and may also allow for comparisons of developmental stages between two different genotypes.
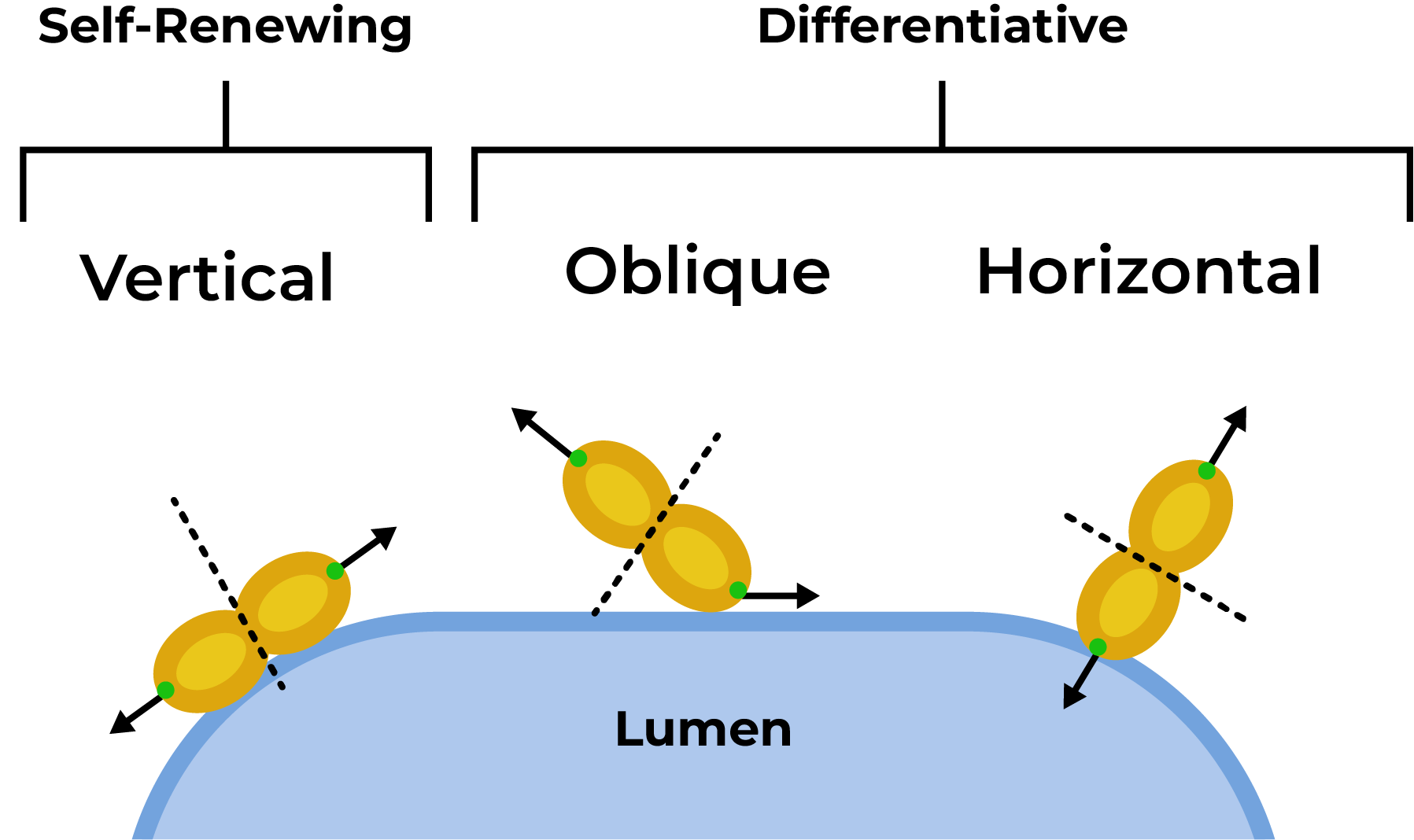
The angle at which the cell divides, relative to the lumen, determines whether the division will result in self-renewal or differentiation. More specifically: a vertical division, with an angle between 60 and 90 degrees from the apical wall to the mitotic spindle, leads to self-renewing divisions. On the other hand, a horizontal or oblique division, with an angle between 0 and 60 degrees, leads to differentiating divisions (Figure 4).
Figure 4: Sketch representing the two modalities of cell division at the level of the rosette lumen: self-renewing and differentiative divisions. This image was adapted from Birtele et al., (https://www.nature.com/articles/s41593-023-01477-3) under the terms and conditions of the Creative Commons Attribution license (CC BY 4.0).
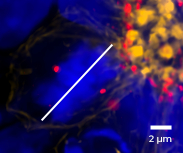
Thanks to X-Light V3 optical sectioning, we were able to image dividing cells close to the lumen and to identify the orientation of the mitosis. Figure 5 (left) shows the lumen of a neural rosette and a cell near it that is dividing. As shown in Figure 5 (right), the angle of cell division is about 90 degrees, suggesting that self-renewing mitosis is taking place.
Figure 5: Left: MIP (1 µm) of the neural rosette’s lumen; Right: crop of dividing cell. The white line shows the orientation of cell division with respect to the lumen. Neural rosettes are stained with: DAPI (blue), Phalloidin (yellow) and Pericentrin (red). Objective: Nikon CFI Plan Apochromat Lambda D 60X oil objective (1.42 NA, 0.15 WD).
Conclusion
In this Application Note, we demonstrated the powerful combination of RosetteArray® plates with the X-Light V3 confocal spinning disk system. Together, these tools offer an advanced solution for neural rosette screening. Additionally, we showed that the X-Light V3 enables high-throughput analysis of neural rosettes, allowing for rapid and efficient assessment of their maturation status.
Acknowledgments
This application note results from a collaboration between CrestOptics, Sapienza University of Rome, and the Center for Life Nano & NeuroScience. We thank Prof. Silvia di Angelantonio (Department of Physiology and Pharmacology, Sapienza University of Rome; Center for Life Nano- & Neuro-Science, Istituto Italiano di Tecnologia; D-Tails srl BC), Dr Lorenza Mautone (Department of Physiology and Pharmacology, Sapienza University of Rome; Center for Life Nano- & Neuro-Science, Istituto Italiano di Tecnologia), Dr. Chiara D’antoni (Department of Physiology and Pharmacology, Sapienza University of Rome; Center for Life Nano- & Neuro-Science, Istituto Italiano di Tecnologia), Dr. Francesca Nistri (Department of Physiology and Pharmacology, Sapienza University of Rome; Center for Life Nano- & Neuro-Science, Istituto Italiano di Tecnologia) for kindly providing the neural rosettes shown in this Application note.
We thank Neurosetta for kindly providing the RosetteArray® plates.